
SugarArray: A User-centred-designed Platform for the Analysis of
Lectin and Glycan Microarrays
Aurora Sucre
1,2 a
, Raquel Pazos
3
, Niels-Christian Reichardt
3,4
and Alba Garín-Muga
1,2 b
1
Biodonostia, eHealth Group, Donostia-San Sebastián 20014, Spain
2
Vicomtech, eHealth and Biomedical Applications, Donostia-San Sebastian 20014, Spain
3
CIC biomaGUNE, Glycotechnology Laboratory, Paseo Miramón 182, 20014, San Sebastián, Spain
4
CIBER-BBN, Paseo Miramón 182, 20014, San Sebastian, Spain
Keywords: Glycomics, Lectin Microarray, Glycan Microarray, Microarray Data Analysis, Data Visualization.
Abstract: Glycan and lectin microarrays are two arising technologies, very important to the glycomics field. Glycomics
is the science that focuses on defining the structures and functions of carbohydrates in nature. These
microarrays provide information regarding the interactions between specific carbohydrates and proteins, and
it has many applications in clinical and research settings. Nevertheless, the availability of analytical software
for these types of arrays is very limited, so researchers usually perform data processing and analytical
pipelines manually, which is very time consuming and prone to error. SugarArray was born as a user-friendly
and intuitive stand-alone solution that process the intensity data generated from glycan or lectin array studies,
and displays the results to the user in an understandable manner. The solution also allows the users to manage
the data as needed, create data plots and automatically generate reports. This tool was intended to simplify
the processing steps of the analytical pipeline, so the users can focus on what really matters: understanding
the results.
1 INTRODUCTION
In recent years, the usage of microarray technologies
in functional glycomics has grown exponentially due
to the great potential of lectin and glycan microarrays
for this field. These types of array provide deep
insight regarding the interactions between glycans
and lectins, useful in multiple clinical and research
settings. For example, they can be used to analyse the
glycosylation profile of glycoconjugates, to perform
quantitative analysis of lectin-glycoprotein
interactions, to discover glycan-related biomarkers in
cancer and to study the cell-surface glycans, among
many other applications (Hu and Wong, 2009).
In this context, the project Glicobiomed was born
as a collaboration between various centres in order to
study the role of glycans in different settings, to
develop new methodologies for glycoanalysis and
ultimately, to obtain novel biomarkers.
To develop a successful project, it is necessary to
have tools that harness the potential of these
a
https://orcid.org/0000-0002-4078-9275
b
https://orcid.org/0000-0002-7160-1191
microarray techniques. Nevertheless, data analysis in
this field has not reach is highest potential due to the
limited availability of analytical software. Even if
some platforms may be available, they do not fulfil
the requirements of the end-users involved in this
project. Therefore, they usually follow a manual
analytical pipeline, which is not desired.
To be able to surpass the limitations associated to
the traditional approach, we have developed a
microarray analysis software called SugarArray,
which provides a solution regarding lectin and glycan
microarray data processing, visualization and
analysis, encapsulated in a user-friendly stand-alone
software. In order to formulate the software
requirements and develop the desired solution, we
followed a user-centred design approach, allowing
the end-users to be involved in every stage of the
development, and assist in the making of a tool that
actually fulfils the needs of the users.
106
Sucre, A., Pazos, R., Reichardt, N. and Garín-Muga, A.
SugarArray: A User-centred-designed Platform for the Analysis of Lectin and Glycan Microarrays.
DOI: 10.5220/0008960101060116
In Proceedings of the 13th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2020) - Volume 5: HEALTHINF, pages 106-116
ISBN: 978-989-758-398-8; ISSN: 2184-4305
Copyright
c
2022 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved

2 RELATED WORK
No tools have been found in the literature that fill all
the gaps in microarray analysis detected by the end-
users involved in the project, as most of the scientific
efforts in this field are put on developing better and
more diverse microarrays instead of developing tools
for analysis.
Almost every commercially available software for
microarray analysis focus on gene or peptide
microarrays, which is not useful for lectin and glycan
analyses. Also, some other tools were detected that do
focus on glycans and lectins, but they analyse data
coming from other technologies, such as mass
spectrometry (Goldberg et. al., 2005; Maass et. al.,
2007), or focus more on the molecules structure and
their representation (Aoki-Kinoshita, 2008), which is
still not the target of this project.
Nevertheless, two software developed specifically
for glycan analysis were detected. The first software
is a stand-alone program composed by a suite of
modules to store, retrieve and display glycan
microarray data. It provides an internal database to
store all the information related to the glycans, their
associated proteins and the experimental data; it also
provides different tools for data visualization, sorting
and filtering; and finally, it includes modules for
automatic plots generation (Stoll and Feizi, 2010).
The second tool is called GLAD (Glycan Array
Dashboard), and it is a web-based tool that provides
functions to visualize and analyse glycan microarray
data, and also compare information coming from
different experiments. It has a module for basic plot
generation (i.e. bar charts) for single-sample data, or
complex plot generation for comparison of samples.
This tool also includes a module for data
normalization between samples, necessary for
comparison (Mehta and Cummings, 2019).
However, none of the described programs fulfil all
the needs of the project, according to the participating
end-users. The main drawback is that the software
interfaces are not as intuitive and easy-to-use as
desired, so the different included functionalities are
not easy to exploit. Another downside is that these
programs were designed strictly for glycan array
analysis, so they do not work with lectin arrays, which
is one of the main requirements. Therefore, the best
approach was to create a tool from scratch and include
all the desired functionalities gradually.
3 LECTIN AND GLYCAN
MICROARRAYS
3.1 Lectins, Glycans and Glycoproteins
Lectins are a group of proteins that present certain
binding behaviour toward carbohydrates, specifically
soluble carbohydrates and the residues of
glycoconjugates (i.e. glycans). These proteins bind
saccharides reversibly and with high specificity, but
can have more than one-binding size along a single
molecule, so they can be specific for more than one
sugar molecule at once.
Lectins can be found commonly in nature: in
plants, animals and bacteria; and they have been
associated with a broad set of functions depending on
where they are found. It is interesting to highlight
their role as recognition molecules in cell-molecule
and cell-cell interactions, affecting a wide range of
cellular events (Lis and Sharon, 1998).
These molecules do not only have a role in nature,
but many different usages have been given to them in
clinical and experimental settings, for example, in
blood typing, histochemical analyses and
biomolecules purification.
Another important group of biomolecules are the
glycans. In general, glycan is considered as a
synonym for polysaccharide, which are “compounds
consisting of a large number of monosaccharides
linked glycosidically” (IUPAC, 1997), usually
formed by more than 10 sugar residues. Nevertheless,
in this context the term will be associated to the
saccharide portion of a glycoconjugate molecule,
which are molecules of carbohydrate bonded to other
compounds (i.e. glycoprotein, glycolipid or
proteoglycan) (Dwek, 1996).
Historically, it was believed that the solely
function of sugar molecules was being a source of
energy, but now it is well-known that they have many
other functions in the biological systems. Glycomics
is the science that focuses on studying the glycome of
the organisms, trying to define structures and
functions of carbohydrates in nature.
Glycoproteins are formed through direct
interactions between glycans and proteins. The
glycans of these molecules can also be attached to
other macromolecules, which indirectly control the
glycoprotein conformation, stability, turnover,
oligomerization and cell surface resident time
(Cummings and Pierce, 2014).
It is important to highlight that the proteins
conforming the glycoproteins are not lectins. Lectins
may temporary bind the sugar fragment of
glycoproteins in order to execute certain functions,
SugarArray: A User-centred-designed Platform for the Analysis of Lectin and Glycan Microarrays
107

but the interaction between the glycan and protein
forming a single glycoprotein is permanent and
formed differently.
Based on how the glycoproteins are joined, they
can be classified as N-linked or O-linked molecules.
N-linked glycoproteins are formed in the
endoplasmic reticulum, where the glycan is attached
to the protein through a nitrogen atom. On the other
hand, O-linked glycoproteins are formed in the Golgi
apparatus, where the linked is created through an
oxygen atom. Then, the newly formed molecules
travel to the plasma membrane, where the saccharide
part is placed facing out (Robb, 2019).
Glycoproteins promote various cellular functions
such as cell adhesion, cell-matrix interactions and
cellular signalling.
It can be expected that abnormalities in the
synthesis of glycoproteins can be associated with
numerous conditions and diseases; therefore, it is
important to understand their mechanisms and
functions in the different organisms.
3.2 Microarray Technologies
Glycan and lectin microarrays are currently
considered two of the most relevant technologies in
functional glycomics, as they help on understanding
the function, interactions and structures of glycans
and glycoconjugates.
A lectin microarray is a functionalized glass plate
with numerous micrometric wells containing
immobilized lectins. As previously described, lectins
have a recognition domain for carbohydrates, so these
panels are very useful in order to study glycans and
glycoproteins. In a microarray experiment, each of
the immobilized lectins can interact with a
specifically fluorescently labelled molecule, thus
generating a characteristic interaction profile for each
glycoconjugate, i.e. it is possible to identify and
measure the glycoconjugates found on a sample based
on the interactions with the lectins on a plate (Hu and
Wong, 2009).
Equivalently, a glycan microarray has wells
containing immobilized glycans instead. These
glycans are able to interact with specifically
fluorescently labelled lectins and help on detecting
the presence and estimating the concentration of
different lectins in a given sample.
Functioning microarrays are created using a
spotter, which is a tool that deposits the different
ligands in the well where they correspond, based on
the experiment design information sheet (GAL file).
Usually, biological replicates are included in the
design in order to obtain more reliable and significant
results; this is done by filling multiple wells with the
same ligand.
In a single microarray plate, it is possible to have
multiple replicated subarrays of ligands, which allow
the researchers to study multiple samples at once with
respect to the same set of ligands. Each sample is
poured over one of the subarrays, so the information
generated on each section of the microarray will
correspond to a single sample. Each subarray is
delimited, so the samples are not mixed up.
3.3 Microarray Data Processing
As previously mentioned, the samples that are going
to be analysed in a microarray should be molecularly
marked somehow. The most common technique is to
use fluorescent molecules, which are bonded to the
molecules in the sample of interest. Then, when these
molecules interact with the ligands in the microarray
wells, the fluorescent molecule is released, and a
fluorescent signal is emitted. By analysing the
emissions of fluorescent signals, the researcher is able
to identify the molecules in the sample and estimate
their concentration, based on the known relations
between glycans and lectins. Depending on the
location of the fluorescent emissions, it is possible to
know which microarray ligands interacted with
molecules of the sample.
In order to obtain information regarding the
fluorescent emissions, a specific scanner is needed,
which captures the emission of light and generates a
monochromatic image (TIFF file) describing the
emissions. Then, different programs can be used to
extract the intensity data, for instance the
ScanExpress, which measures fluorescence intensity
at each point, recognises different patterns, makes
certain corrections and adapts the measurements to
the array design according to the GAL file finally
generates a CSV file describing the intensities
associated to each microarray spot and various
associated statistics.
3.4 Microarray Data Analysis
Traditionally, researchers read and analyse the
intensities CSV files using a spreadsheet software
such as Microsoft Excel. This approach allows them
to create data charts and basic plots to visualize and
understand the data, but managing large amounts of
data and stablishing comparison protocols is not so
easy due to limitations of spreadsheet software. Also,
this approach is prone to misleading results, due to the
multiple variables that are manually controlled and
can lead to erroneous calculations. Finally, this
HEALTHINF 2020 - 13th International Conference on Health Informatics
108

approach is very time consuming because every
calculation and chart is created manually; the whole
analytical process should be supervised by the
researchers.
Based on these observations it became interesting
to develop a tool that automatizes the analytical
pipeline and enables the users to visualize the data
and generate reports on a fraction of the time. This
process assures reliable results, as they are processed
automatically, not relying in manually performed
tasks that are prone to errors.
4 SugarArray
The SugarArray solution is a stand-alone software
that was developed in order to analyse the intensity
data generated after scanning lectin and glycan
microarrays. SugarArray processes the data and
generates various types of plots so that it is possible
to analyse the data in depth and generate reports in an
easy and visual manner.
The software was developed following a user-
centred design approach and consists of a graphical
user interface (GUI) designed using QT, and
functionalized using Python; and a set of analytical
modules, also developed in Python.
4.1 Software Design Approach
The user-centred design (UCD) approach is an
iterative design process where the end-users are
involved in all the stages of design and development.
This approach enables to better describe the user
needs and help the developers while defining the
solutions to the user-detected problems. Different
methods were considered along the process, but they
can be sorted in 2 categories: investigative and
generative methods. The first category comprises the
techniques that allowed us to understand the context
of the problem and to better define the user needs,
while the second set of techniques allow the users to
present their requirements for the software and their
ideas that may help developers achieving the project
goals (Nugraha and Benyon, 2010).
As previously stated, the development of this
software was embedded within a larger glycomics
project, so a team of end-users was available in order
to follow this approach properly. This team consisted
of five glycomics researchers which were actively
involved in the design process along with the
developers. The number of involved researchers was
chosen following Jakob Nielsen’s recommendations
regarding usability testing (Nielsen, 1993; Nielsen,
2000).
The workflow of the followed UCD methodology
is represented in Figure 1.
Figure 1: User-centred design methodology workflow.
The initial design step consisted on having a face-
to-face meeting between developers and two of the
chosen end-users where we used a storytelling tool so
the users could help developers understand their
current analytical practices and protocols, the data
they usually exploit, the results they obtain and the
general context of the project. They also provide
information regarding their initial expectations with
respect to the new analytical tool, and described their
dissatisfaction about the commercial tools available.
The design team prepared an initial basic version
of the software, trying to incorporate the different
elements needed to replicate the behaviour of their
current practices, but in a simpler and automated
manner.
All the involved end-users tested this first version
of the software and were all pleased with the initial
results. Afterwards, the design process consisted on
having periodically face-to-face meetings where a
brainstorming approach was followed so both, the
end-users and developers, define together further
requirements of the tool, interesting functionalities to
incorporate and the expected looks of the application.
After each brainstorming session, the developers
modify the software in order to fulfil the newly
appearing requirements and present each new version
to the end-users for feedback, who evaluated the tool
focusing on its usability and effectiveness. This cycle
continued until a fully-functional tool that fulfil all
the user requirements was developed.
SugarArray: A User-centred-designed Platform for the Analysis of Lectin and Glycan Microarrays
109
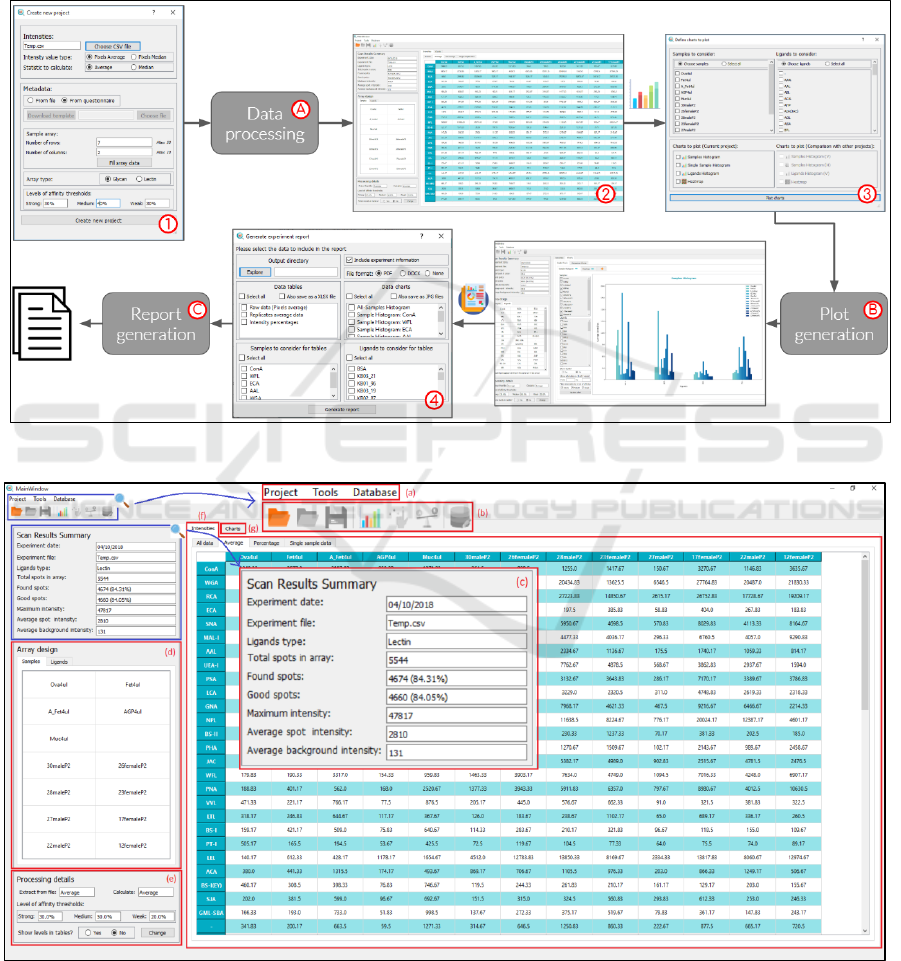
4.2 Analytical Pipeline
In order to perform a complete analysis on the data
generated from a microarray experiment the steps shown in
Figure 2 are followed. This pipeline represents the
interfaces through which the user interacts with the
software, and the functional modules that are executed after
each user’s action.
As shown in Figure 2, the GUI comprises 4 dialogs: (1)
the new project dialog , (2) the main window, (3) the charts
dialog and (4) the report dialog.
On the other hand, the set of analytical tools
includes 3 main modules for: (A) data processing, (B)
plot generation and (C) report generation.
Figure 2: SugarArray analytical pipeline.
Figure 3: GUI - Main window: (a) Menu bar, (b) Tool bar, (c) Scanner metadata section, (d) Array design section, (e)
Processing details section, (f) Data tables section and (g) Data charts section.
HEALTHINF 2020 - 13th International Conference on Health Informatics
110

All the mentioned dialogs and modules will be
shown and described in detail in following sections.
4.3 Graphical Interface
4.3.1 Main Window
The main window corresponds to the primary point
of interaction between the user and the analytical
software. It allows the user to start new analysis,
visualize and manage the data and generate and view
charts. The interface is shown in Figure 3 and all its
composing elements will be described next.
The upper section of the window contains two
independent bars: (a) the menu bar, where the
different software actions are sorted in menus; (b) and
the tool bar, where the same actions are presented, but
in this case, using icons. This disposition allows the
user to perform the analytical actions in the way that
is more intuitive for each.
Then, on the left side of the window we have three
elements: (c) the scanner metadata section, where all
the information regarding the capture step is
summarized (experiment date, experiment data file,
total spots in the array, number of found spots,
number of good spots, maximum detected intensity,
average spot intensity and average background
intensity) to provide certain insight on the performed
experiment; (d) the array design section, where two
data tabs are contained, one describing the disposition
of samples within the whole experiment array and the
other describing the disposition of the ligands within
each of the subarrays; and finally, (e) the processing
details section, where the statistics chosen to be
extracted and calculated are shown, along with the
level of affinity thresholds modifiable by the user.
On the right side of the main window we have (f)
a data tab containing various tables describing the
intensity data, and (g) another tab where all the
generated charts will be displayed.
The intensities data section contains 4 tables in 4
tabs: (1) one displaying the data extracted from the
intensities file in a table having as many rows as
ligands and as many columns as samples, (2) another
table showing the average/median values calculated
between the replicates of the same ligand found
within the array, (3) the third one displays the same
calculated values but in terms of percentages, and (4)
the final table shows the averages/medians and
percentages associated to a single sample as chosen
by the user. The latter allows the user to select which
sample to display and how the data will be sorted to
analyse each sample in depth (Figure 4).
Finally, the charts tab is filled-up on the fly as the
user creates new plots. Each newly created chart will
be displayed in an independent tab within the charts
section. Each tab has a set of tools that allow the user
to modify the charts interactively after creation, by
modifying the ligands/samples included in the charts
and how the information is sorted, deciding whether
the labels are shown, etc. (Figure 5).
Figure 4: GUI - Main window (Sample view).
Figure 5: GUI - Main window (Chart view).
4.3.2 New Project Dialog
The new project dialog allows the user to input the
files and data necessary to define a new experiment in
the platform. The interface is shown in Figure 6.
On top of the dialog we have (a) the intensities
section, where the user must select the intensities file
generated by the scanner software and define which
information to extract from it and which statistic to
calculate between replicates.
SugarArray: A User-centred-designed Platform for the Analysis of Lectin and Glycan Microarrays
111
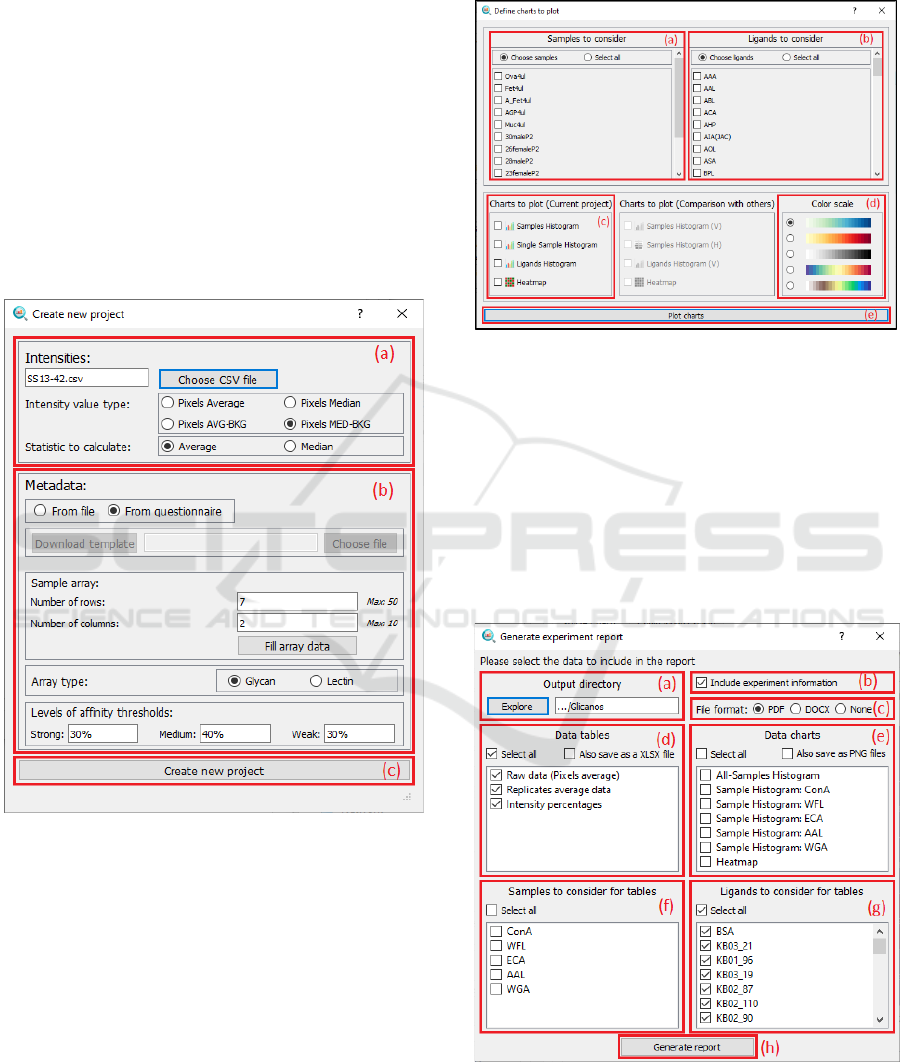
Then, on the middle of the dialog (b) the metadata
section is shown. This section allows the user to
provide further information needed for the analysis.
The user is able to (1) upload a metadata file or (2)
define the metadata in a questionnaire. For the first
option, a data template must be followed. For the
second option, different widgets are displayed so the
user can provide all the metadata values easily. First,
the user must define the sample array disposition and
then write down the names of the samples in the array.
Then, the user must define the type of array and the
percentages associated to the levels of affinity to
classify the sample-ligand interactions.
Finally, in the bottom of the dialog (c) the “create
new project” button is shown. When this button is
clicked , the data processing pipeline starts.
Figure 6: GUI - New project window: (a) Intensities section,
(b) Metadata section and (c) Create button.
4.3.3 Charts Dialog
The charts dialog allows the user to define the different
charts that are needed to analyse the data in depth. The
interface is shown in Figure 7.
On the top section of the window we have two
elements: (a) a list widget that allow the user to select
the samples that must be included in the charts, and (b)
another list widget, in this case to select ligands.
On the centre of the dialog we have (c) a list of
check boxes, so the users can choose the plots to draw;
and (d) a list of colour scales so the user can define the
appearance of the charts.
Finally, in the bottom of the window we have (e)
the “plot charts” button, so when it is clicked, the plots
are created.
Figure 7: GUI - Charts dialog: (a) Samples list, (b) Ligands
list, (c) Charts list, (d) Colour scale list and (e) Plot button.
4.3.4 Report dialog
The report dialog allows the user to generate reports
and define which information to show in them. It is
possible to generate full reports in PDF or DOCX
format, but also generate data table reports (XLSX
files) or export the generated plots for further use.
The interface associated to this report wizard is
shown in Figure 8.
Figure 8: GUI - Report dialog: (a) File explorer section, (b)
Information checkbox, (c) Format section, (d) Tables section,
(e) Charts section, (f) Tables-Samples section, (g) Tables-
Ligands section and (h) Generate button.
HEALTHINF 2020 - 13th International Conference on Health Informatics
112

On the top of the dialog we have (a) the file
explorer section, where the user must define where to
store the files that will be generated, (b) a checkbox to
decide whether or not to include the experiment
information, and (c) a list to define the output format.
In the middle of the dialog we have 2 lists: (d) one
allows the user to select the data tables to include in the
report and (e) the other one, to decide which plots to
include.
Finally, in the bottom of the dialog we have another
set of lists and a “Generate report” button (h). The latter
set of lists allow the user to select the samples (f) and
the ligands (g) to be included in the tables.
4.4 Analytical Modules
4.4.1 Microarray Data Processing
The data processing module of the software is
composed of two analytical sub-modules:
GetMetadata and GetValues module. These modules
include a variety of methods that are followed in order
to process the data, extract the relevant information and
calculate statistics; these methods are mainly based on
well-known Python packages for data management
and statistics: NumPy and Pandas.
The GetMetadata sub-module comprises the
functions necessary to extract the metadata information
from a text file if it was provided. Otherwise, the
metadata information is directly obtained from the
“New Project” interface.
First, the software checks if the provided file has
the expected format and if so, extracts all the data and
stores it in such a way that can be exploited by the
software and easily shown in the main window later.
On the other hand, the GetValues sub-module
contains all the functions needed for intensity data
extraction and processing.
The first step is to read the intensities file, extract
the metadata associated to the scanner, detect the file
column where the chosen-statistic values are stored,
and save the selected raw data in a dataframe. This
initial dataframe contains a list of items, where each
item contains the coordinates and intensity value
associated to each array spot. However, this data
structure is still not compatible with the software.
Then, a series of functions are performed on the
raw data to extract the ligands information and modify
the data structure based on that. In particular, we detect
how many replicates are found for each ligand and
define a dataframe describing how the different ligands
were distributed in the microarray; we also assure that
independent non-replica ligands are named differently;
and finally, after a series of concatenated
transformations, we obtain a dataframe having as many
rows as ligands in and as many columns as samples,
assuring that the data associated to non-existing ligands
or samples is excluded.
Once the data is structured in a ready-to-show
manner, the next step is to calculate the chosen statistic
(average or median) between the replicates of the same
ligand. The obtained dataframe has the one row for
each ligand (instead of the n replicates that were shown
before).
Next, we calculate the percentage associated to
each calculated statistic. Each percentage represents
the ratio of intensity of each ligand, in comparison to
the maximum intensity associated to each sample.
Finally, we must define the “levels” dataframe
based on the given values associated to each affinity
threshold and on the calculated statistics dataframe.
We must state which is the affinity level associated to
each ligand for each sample. The affinity percentages
describe which number of ligands can be considered as
having a high/moderate/weak interaction with each
studied sample. The default values defined by
glycomics experts involved in the project state that the
30% of samples having a higher intensity value can be
associated to strong interactions, the next 50% with
moderate interactions and the bottom 20% of the
ligands, a weak interaction with the sample. The
percentages can be modified as desired, and so the
number of ligands defined by each category. A
dataframe is returned, describing the level associated
to each position in the matrix.
The GetValues sub-module may also work
following a shortened path, where only the levels
dataframe is recalculated based on modifications made
by the user in the main window.
4.4.2 Plots Generation
This analytical module comprises the functions needed
for plot generation and edition. The methods followed
to create the different plots are based on functions from
a set of well-known Python packages for data
visualization: matplotlib and seaborn.
Currently the tool allows the users to create four
types of charts, shown in Figure 9.
The samples histogram describes the intensity data
for all the chosen ligands regarding all the chosen
samples. The chosen ligands are represented in the X-
axis, the intensities in the Y-axis and a different colour
is assigned to each sample. Therefore, each shown bar
represents the mean/median intensity for a ligand
regarding a sample. The single sample histogram is
similar, but in this case, only the intensity values for
SugarArray: A User-centred-designed Platform for the Analysis of Lectin and Glycan Microarrays
113
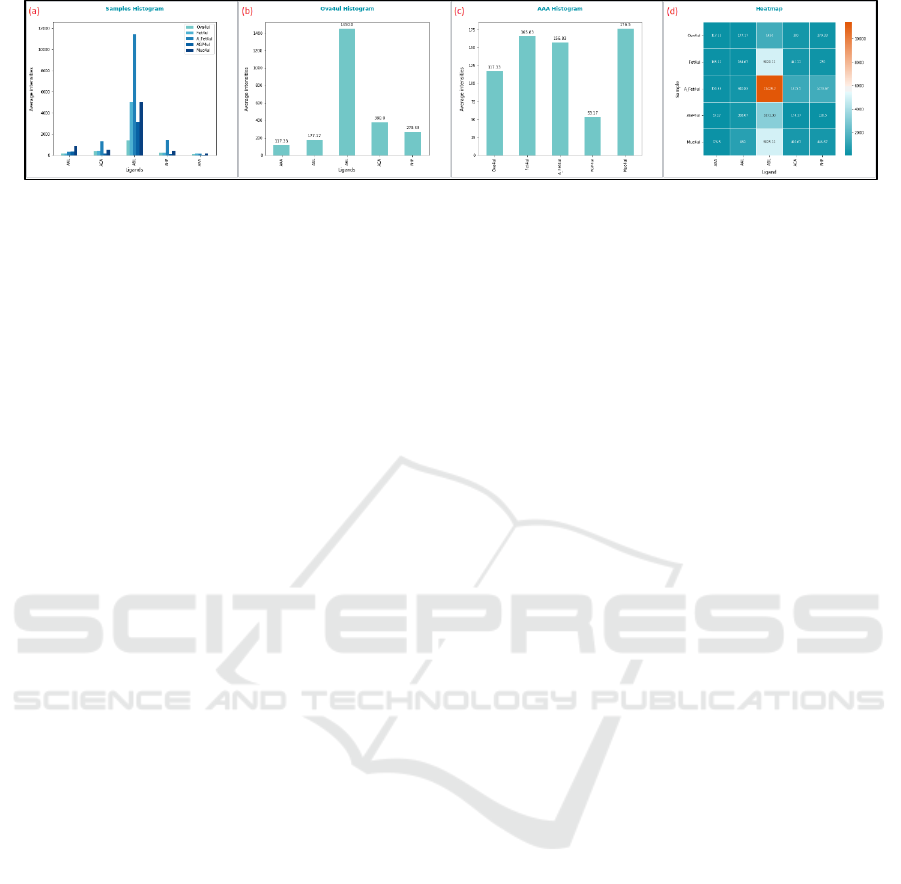
Figure 9: Generated plots: (a) Samples histogram, (b) Single sample histogram, (c) Ligand histogram and (d) Heatmap.
one single sample are represented and thus, one bar per
ligand is shown.
On the other hand, the ligand histogram represents
the chosen samples in the X-axis and draws the bars
represented the mean/median intensity detected on
each sample for a single chosen ligand.
Finally, the heatmap is a colour matrix having as
many rows as chosen samples and as many columns as
chosen ligands, where each cell is coloured based on
the intensity value associated to each ligand for each
sample, following a colormap.
The module creates the plots based on the initial
choices of the user, but can also modify them based on
the interactions between the user and the interface.
Every time the user changes something in a chart
view on the main window, such as add/remove
ligands/samples, show/hide labels, filter based on
values or filter by level of affinity a new plot is created
to substitute the previous one that was shown to the
user. The only non-modifiable feature is the colour
scale, so the one chosen at first is kept.
4.4.3 Generation of Reports
The report-generation module allows the user to export
the processed data and the generated charts when
desired, providing certain flexibility in order to fulfil
all the requirements defined by the end-users.
Currently, it is possible to generate a full report in PDF
or DOCX format. It is possible to include different sets
of information on each generated report, choosing
between (1) the experiment data (scanner-provided
information, samples array and ligands array), (2) the
data tables (raw, statistics or percentages tables) or (3)
the generated plots. The user can decide to include all
data tables/plots or just a selection of them.
Regarding table generation, the user is also able
to decide which samples and ligands to include.
Finally, it is also possible to export the generated
tables independently in a format that is easier to
manage (XLSX), and also to save the plots as images
(PNG).
When generating a report, the users are able to
select between these options the ones that suit them
better. The software will generate a set of files based
on the user choices and stored them all in the selected
location.
Just like the plots, the reports can be generated
iteratively as needed, in order to include different sets
of information on each, without the risk of losing the
ignored experiment data.
5 DISCUSSION
In this paper we proposed a microarray analysis
stand-alone software that is able to study both lectin
and glycan microarrays, and we also describe the
user-centred-design approach that was followed in
order to develop a tool aligned with the real
necessities of real users.
After having analysed the different commercial
tools available we did not find a tool that was
compatible with both types of arrays nor included the
analytical functionalities that were desired for the
software, so a new stand-alone program was created
from scratch.
This newly-developed software allows the end-
users to simplify their analytical pipeline, execute all
the tedious tasks automatically and without mistakes
(which cannot be assured when executing manual
processing), and focus on result interpretation steps
rather than on data processing itself.
SugarArray allows researchers to input the raw
analytical data, obtain tables and plots in just a few
steps, and exporting the generated information in
different formats for further analysis, or for report
generation.
A proper validation process was not conceived at
this stage of the project, because the UCD approach
that was followed allowed us to have constant testing
and feedback from end-users regarding the software
behaviour. Nevertheless, a pre-validation process was
conducted by the end-users in order to test the final
functional version of the software. Also, the
development team is currently defining the protocols
HEALTHINF 2020 - 13th International Conference on Health Informatics
114

that will be followed in the future in order to properly
validate the software.
The pre-validation analysis consisted of two main
tasks: (1) evaluate the behaviour of the different
modules and the interaction between the user and the
software and (2) compare the analytical results
obtained using the SugarArray tool with those
obtained following the manual approach.
The feedback that has been given so far has been
positive. The comparison between SugarArray results
and manually obtained results was favourable; every
experiment that was involved in the comparison
returned the same results for both approaches.
Concerning software usability, the users’ opinions
were also optimistic; all the required functionalities
were included and the definition of the analytical
pipeline within the software was simple and easy-to-
understand. However, some minor visual
improvements have been identified, and they will be
considered in following iterations of development.
6 CONCLUSIONS
The chosen user-centred-design methodology
allowed the developers to successfully capture the
end-user necessities and develop a solution that
appears to be appreciated by the end-user.
The development of this tool was embedded
within a larger glycomics project associated to
complex processes and analysis. Therefore, having a
tool that automatizes the data analysis steps is
important to allow researchers to focus on the
meaning of the analytical results, accelerating the
obtention of clinically significant insight.
This functional version of the software is still on
its validation phase. As mentioned, initial pre-
validation steps were followed in order to receive
early feedback from the involved users regarding
their impressions of the software. This initial
feedback was positive, but further insight obtained on
a structured manner, and coming from project-related
and external end-users is needed.
Currently, the validation protocols are being
defined in order to obtain unbiased reviews. These
protocols will consider (1) software usability, (2)
results accuracy, (3) software efficiency and (4)
comparison with other software or approaches.
Besides software validation and the already-
detected visual improvements, the future work will be
focused in the development of new functionalities
sorted in three development lines: Data annotation,
data comparison and outlier management.
For data annotation we are willing to build a
database to store information regarding ligands
classification, interactions, structures, etc. The
software will also include the tools to manage the
database intuitively, and other modules to annotate
the ligands based on the database-stored information.
For data comparison, we need to develop tools for
data normalization in order to make the information
associated to different studies comparable; we are
also willing to expand the plot-generation module in
order to incorporate data from different studies and
generate new tools for comparison.
Finally, the necessity for an outlier management
tool has arisen, that allow the users to detect those
values that do not behave as expected and handle
them in such ways that do not affect the analytical
results.
ACKNOWLEDGEMENTS
This work has been funded by the Basque
Government by means of the ELKARTEK program
within the context of the Glicobiomed (BMG18)
project: “Tools and Opportunities for Biomedical
Glycoscience”.
We would also like to offer our special thanks to
the CIC biomaGUNE Glycotechnology Lab team for
their participation in the project as the end-users
committee involved in the user-centred design
process.
REFERENCES
Aoki-Kinoshita, K. (2008) An introduction to
bioinformatics for glycomics research. PLoS.
Computational Biology 4(5):e1000075. doi:
10.1371/journal.pcbi.1000075.
Cummings, R. and Pierce, J. (2014) The Challenge and
Promise of Glycomics. Chemistry and Biology 21(1):1-
15. doi: 10.1016/j.chembiol.2013.12.010
Dwek R. (1996). Glycobiology: Toward Understanding the
Function of Sugars. Chemical Reviews 96(2): 683-720.
doi: 10.1021/cr940283b
Goldberg, D., Sutton-Smith, M., Paulson, J. and Dell, A.
(2005). Automatic annotation of matrix-assisted laser
desorption/ionization N-glycan spectra. Proteomics
5(4):865 – 875. doi: 10.1002/pmic.200401071.
Hu, S. and Wong, D. (2009) Lectin microarray. Proteomics
Clin Appl. 3(2):148-154. doi: 10.1002/prca.200800153
IUPAC (1997). Compendium of Chemical Terminology.
Compiled by A. D. McNaught and A. Wilkinson.
Blackwell Scientific Publications, Oxford (1997).
SugarArray: A User-centred-designed Platform for the Analysis of Lectin and Glycan Microarrays
115

Online version (2019-) created by S. J. Chalk. doi:
10.1351/goldbook.
Lis, H. and Sharon, N. (1998). Lectins: Carbohydrate-
Specific Proteins That Mediate Cellular Recognition.
Chemical Reviews 98(2):637-674. doi:
10.1021/cr940413g
Maass, K., Ranzinger, R., Geyer, H., von der Lieth, C. and
Geyer, R. (2007) “Glyco-peakfinder” – de novo
composition analysis of glycoconjugates. Proteomics
7(24):4435 – 4444. doi: 10.1002/pmic.200700253.
Mehta A. and Cummings R. (2019). GLAD: GLycan Array
Dashboard, a visual analytics tool for glycan
microarrays. Bioinformatics 35(18): 3536-3537. doi:
10.1093/bioinformatics/btz075
Nielsen, J. (1993). Usability engineering. Academic Press,
Boston (1993).
Nielsen, J. (2000). Why you only need to test with 5 users?
Nielsen Norman Group. Available at:
www.nngroup.com/articles/why-you-only-need-to-
test-with-5-users [Accessed December 11, 2019]
Nugraha, S. and Benyon, D. (2010). Designing Interactive
Systems: A comprehensive guide to HCI and
interaction design. Pearson Education Limited. Harlow,
2nd edition.
Robb, A. (2019). What Are Glycoproteins? - Definition,
Functions & Examples. Study.com. Available at:
study.com/academy/lesson/what-are-glycoproteins-
definition-functions-examples.html [Accessed October
22, 2019]
Stoll M. and Feizi T. (2010). Software Tools for Storing,
Processing and Displaying Carbohydrate Microarray
Data. In Glyco-Bioinformatics – Bits ‘n Bytes of Sugars
Proceedings. BEILSTEIN INSTITUT.
HEALTHINF 2020 - 13th International Conference on Health Informatics
116
