
AdoBPRIM: Towards a New Healthcare Risk-aware Business Process
Management Tool
Rafika Thabet
1,2
, Amine Boufaied
1
, Elyes Lamine
3
, Dominik Bork
4 a
, Ouajdi Korbaa
1 b
and Herv
´
e Pingaud
2
1
University of Sousse, ISITCom, MARS Research Lab, Route G.P.1, 4011, Hammam Sousse, Tunisia
2
University of Toulouse, INP, CNRS, LGC, F-31432 Toulouse Cedex 04, France
3
University of Toulouse, IMT Mines Albi, Department of Industrial Engineering, Route de Teillet, 81013 Albi Cedex 9, France
4
University of Vienna, Faculty of Computer Science, W
¨
ahringer Street 29, 1090 Vienna, Austria
Keywords:
Medication Use Process, Medication Error, Risk-aware Business Process Management, BPRIM, ADOxx.
Abstract:
Performing risk management in healthcare facilities is particularly difficult due to the highly dynamic, complex,
and multi-disciplinary nature of healthcare processes like the Medication Use Process (MUP). Risk-aware
Business Process Management (R-BPM) is a promising approach to obtain a better understanding of such
processes by identifying and analyzing corresponding risks. However, not all R-BPM approaches perform
well in capturing the complexity of clinical processes. In this work, we introduce a new R-BPM framework
called BPRIM that allows the identification and the analysis of medication error risks related to the complex
medication use process. BPRIM is implemented using the ADOxx meta-modelling platform and then tested in
a real case study. The tool is specific to the case study, but the framework can be used also in other healthcare
processes.
1 INTRODUCTION
Patient safety is a priority for several government
agencies, non-profit organizations, and regulatory bod-
ies considering the detrimental and financial conse-
quences associated with Medication Errors (MEs) and
Adverse Drug Events (ADEs). A ME is an unintended
failure in the Medication Use Process (MUP) that leads
to, or has the potential to lead to, ADE for the patient.
Indeed, MUP is a complex and multidisciplinary pro-
cess that involves numerous professionals and com-
poses several steps. This complexity causes the occur-
rence of MEs, which can lead to serious health-related
consequences for the patients. In 2015, the French Na-
tional Authority for Health considered that 40% of the
serious adverse events are of medication origin (HAS,
2013).
Many international studies and reports recognized
that the majority of errors were not the result of reck-
less behavior on the part of healthcare providers, but
occurred as a result of the speed and complexity of
a
https://orcid.org/0000-0001-8259-2297
b
https://orcid.org/0000-0003-4462-1805
the MUP. Reports concluded that many patients die
during hospital admission as well as during primary
care, simply due to medication errors (Moyen et al.,
2008). Most of these errors are not coincidental but
systematic in origin. Although most available studies
originate from the USA, studies conducted in other
countries such as the United Kingdom (Leape et al.,
1991) and the Netherlands (Barker et al., 2002) have
shown similar results. This strongly implies that med-
ication errors are a major public health problem in
many western as well as developing countries.
Reducing the MEs risk is a shared responsibility
between patients, healthcare professionals, regulators,
and the pharmaceutical industry at all levels of health-
care delivery. (Kane-Gill et al., 2017) recognizes that
preventing harm resulting from adverse drug events
may be circumvented by averting medication errors
with appropriate risk management approaches.
What needs to change is the way in which health-
care risk management methods are orientated. Indeed,
in the healthcare field, several risk management meth-
ods exist (HAS, 2013). However, these methods do
not consider the complexity of MEs, its strong con-
498
Thabet, R., Boufaied, A., Lamine, E., Bork, D., Korbaa, O. and Pingaud, H.
AdoBPRIM: Towards a New Healthcare Risk-aware Business Process Management Tool.
DOI: 10.5220/0008977604980505
In Proceedings of the 13th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2020) - Volume 5: HEALTHINF, pages 498-505
ISBN: 978-989-758-398-8; ISSN: 2184-4305
Copyright
c
2022 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
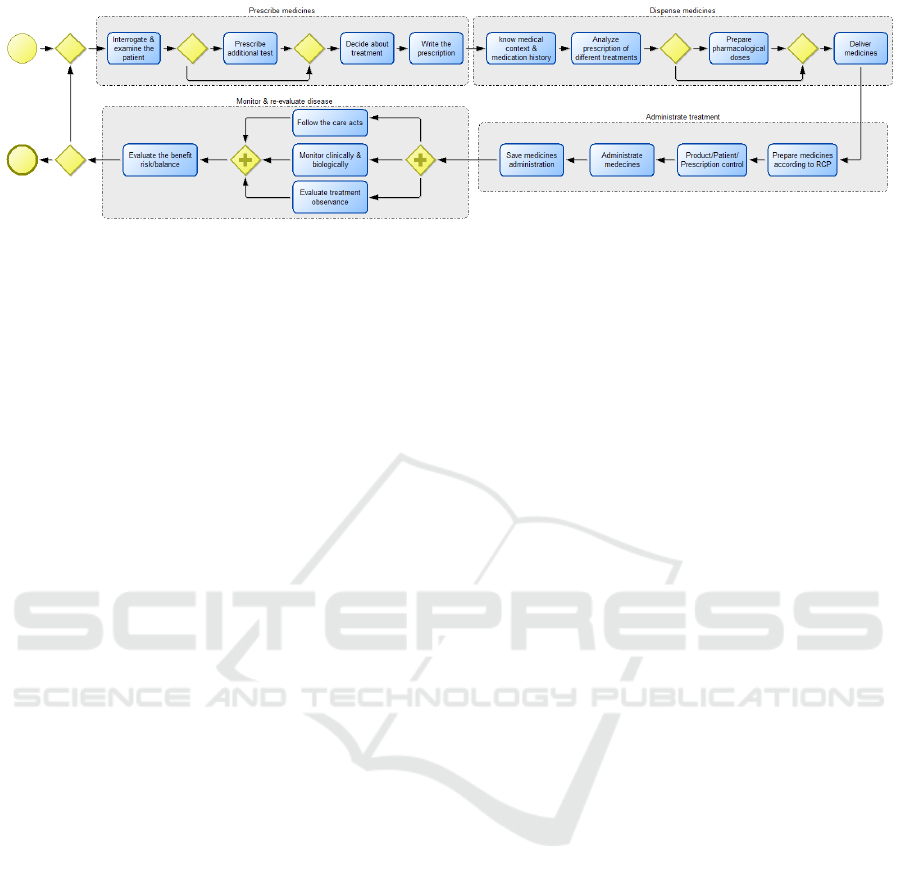
Figure 1: Medication Use Process as BPMN model (based on the French National Authority for Health report (HAS, 2013)).
nection to the MUP activities, and the influence of
organizational and human factors. Following these
observations, it would be essential for the healthcare
facilities to explore new risk management approaches
which consider all related concepts to the ME risk oc-
currence as well as their dependencies inherent to the
steps and activities of the MUP.
To alleviate the shortcoming of existing ap-
proaches, we propose studying the potential of
the Business Process-Risk Management - Integrated
Method (BPRIM) (Lamine et al., 2019; Thabet et al.,
2018) to manage MEs risks related to the com-
plex MUP. BPRIM is an integrated management ap-
proach of risk and business processes following the
Risk-aware Business Process Management (R-BPM)
paradigm (Suriadi et al., 2014; Lamine et al., 2019;
Thabet et al., 2018). R-BPM aims to integrate the
traditionally separated domains of risk management
and business process management. This integration
allows increasing efficiency of the risk identification,
detection, and assessment in relation to the business
processes.
This paper is structured as follows. First, Section 2
provides an overview of the MUP and the impact and
the frequency of the ME occurrence in this process.
In Section 3, we propose a comparative study of the
current risk management methods in healthcare. Our
adopted framework and methodology are presented in
Section 4. An overview of the first results obtained
after the implementation of our framework is given in
Section 5. Finally, Section 6 concludes the paper and
outlines future perspectives.
2 BACKGROUND
In (Vest et al., 2019), the Medication Use Process is
defined as “the fundamental system that provides the
basis for safe medication use within the healthcare en-
vironment.” Thus, ensuring medications are used and
secured in the most appropriate manner and across all
settings. The MUP consists of a complex and multi-
disciplinary process, involving numerous practitioners
and several major steps (i.e., prescribing, dispensing,
administration and medication monitoring).
Figure 1 represents the macro-steps and related ac-
tivities of the MUP as defined in the French National
Authority for Health report (HAS, 2013) by means of a
Business Process Model and Notation (BPMN) model.
Obviously, these activities do not reflect the reality.
In reality, the MUP may involve up to 46 activities
from the moment a doctor considers prescribing medi-
cation to the moment when this medication is actually
administered or taken by the patient.
The complexity of this process increases the likeli-
hood and number of occurrences of Medication Error
(ME) risks. There is no consensus about the defini-
tion of a medication error. A systematic literature
review found 26 different terminologies employed for
a Medication Error (Lisby et al., 2010). As defined
by the United States National Coordinating Council
for Medication Error Reporting and Prevention (NCC
MERP) (NCC MERP and Prevention, 2019), a medi-
cation error is: “any preventable event that may cause
or lead to inappropriate medication use or patient harm
while the medication is in the control of the health care
professional, patient, or consumer. Such events may
be related to professional practice, health care prod-
ucts, procedures, and systems, including prescribing,
order communication, product labelling, packaging,
and nomenclature, compounding, dispensing, distribu-
tion, administration, education, monitoring, and use”.
This definition is broad and suggests that errors are
preventable at different levels.
These risks can be at the origin of Adverse Drug
Event (ADE) with potentially severe clinical conse-
quences for the patient. ADEs were defined as injuries
resulting from medical interventions related to a drug.
Adverse drug events may result from medication errors
or from adverse drug reactions in which there was no
error (Bates et al., 1995). ADEs consequences can be
substantial, including hospital admission, prolonged
hospital stay, additional resource utilization, and lower
AdoBPRIM: Towards a New Healthcare Risk-aware Business Process Management Tool
499

patient satisfaction (Caporossi et al., 2014). In this con-
text, many publications have described the frequency
and the cost of MEs and ADEs in the MUP (Bates,
2007). Starting in 2000, the Institute of Medicine be-
gan publishing a series of reports on quality in health-
care. The first one, “To Err is Human”, increased
the awareness of unacceptably high rates of MEs and
sentinel events in U.S. hospitals with about 44,000 –
98,000 deaths annually attributed to these preventable
mishaps (Committee on Quality of Health Care in
America and Institute of Medicine, 2000). The sec-
ond one, “Crossing the Quality Chasm”, made four
major points: errors are common and costly; systems
cause errors; errors can be prevented and safety can
be improved; and medication-related adverse events
are the single leading cause of injury (Committee on
Quality of Health Care in America and Institute of
Medicine Staff, 2001). The most recent one, “Pre-
venting Medication Errors”, attempts to think about
what needs to be done to reach the next level of med-
ication safety (Committee on Quality of Health Care
in America and Institute of Medicine, 2006). In this
work, first, an epidemiological review showed that, es-
timated very conservatively, medications harm at least
1.5 million people per year. In hospitals, there are at
least 400,000 preventable adverse drug events per year,
or approximately one medication error per patient per
day. That finding probably had the greatest impact on
the general public (Committee on Quality of Health
Care in America and Institute of Medicine, 2006).
The occurrence of these errors questions the ro-
bustness of the management methods actually used to
secure the MUP in the healthcare facilities especially
that knowledge and practices evolve and the organiza-
tion must remain vigilant.
Facing the high frequency and impact of MEs and
ADEs, healthcare facilities have to set up a risk man-
agement approach aiming to ensure patient safety, and
specifically, to decrease ME occurrence related to the
complex MUP. Section 3 provides an overview of some
existing healthcare risk management methods.
3 RISK MANAGEMENT IN
HEALTHCARE FACILITIES
In France, ten years ago, Experience Feedback Com-
mittees (EFCs), were created to analyze ADEs within
a medical ward (Francois et al., 2013). An EFC is a
multidisciplinary team representing the diversity of the
functions encountered in the medical ward. The EFC
members meet regularly to examine reported ADEs
related to their medical ward. They prioritize ADEs
either on the gravity of the event or on the frequency of
occurrence and propose corrective actions. To analyze
the ADE facts, the committee uses one or many of
the existing risk management methods in healthcare
facilities.
A comparative study of the most relevant risk as-
sessment methods currently used in the healthcare do-
main is summarized in Table 1. This comparison is
carried out in accordance with the following criteria:
• Process/Activity based:
whether the method de-
scribes in which system process or activity the
causes occurred;
• Reactive/Proactive:
whether the method is reac-
tive (risk considered ex post) or proactive (ex ante
consideration of risk);
• Qualitative/Quantitative:
whether the method
follows qualitative and/or quantitative analysis;
• Input:
the reasons for which the method has to
start being applied;
• Causes Ranking:
whether the method classifies
different causes;
• Logical Relationships:
whether the method de-
scribes relationships between multiple causes us-
ing logical operators to describe risk scenarios;
• Causes Chronology:
whether the method pro-
vides a chronological reconstruction of causes;
• Causes Description:
which description type is
used to represent causes.
Generally, healthcare risk management methods
can be categorized according to the used risk assess-
ment type: Reactive risk assessment and Proactive
risk assessment (see Table 1). Reactive risk assess-
ment methods are Root Cause Analysis methods based
on systematic questioning to identify the underlying
causes of adverse occurrences.
According to Table 1, only few methods (three
out of 10) tried to integrate system process or activ-
ity where the causes occurred in the analysis process.
However, among these approaches, none provides a
graphical description to represent system activities and
relationships between multiple risk causes using logi-
cal operators. In fact, working with graphs allows to
capture a large analysis scope.
In an organisation, all presented methods will set
up brainstorming meetings with a team of healthcare
providers, as well as quality/risk managers, to reduce
the number of MEs and ADEs. However, from our
point of view, none of these methods is able to satisfy
all the following requirements:
•
Deal with the complex MUP at the correct level by
describing all related and collaborative activities
that compose the process (Rq1);
HEALTHINF 2020 - 13th International Conference on Health Informatics
500

Table 1: Comparing current healthcare risk assessment methods.
Method
Process/
Activity based
Reactive
Proactive
Qualitative
Quantitative
Input
Causes
ranking
Logical
relationships
Causes
chronology
Causes
description
Five Whys ? 2 2 2 2 2
Questioning
2 2 2
Schema/
Table
Ishikawa 2 2 2 2 2
Questioning
2 2 2 Graph
Human Factor Analysis and
Classification System (HFACTS)
2 2 2 2 2
Questioning
2 2 2
Graph/
Grid
Association of Litigation And Risk
Management (ALARM)
2 2 2 2 2
Questioning
2 2 2
Form/
Report
Causal Tree Analysis (CTA) 2 2 2 2 2
Questioning
2 2 2 Tree
ORION 2 2 2 2 2
Questioning
2 2 2
Table/
Report
Failure, mode, effects, and criticality
analysis (FMECA)
2 2 2 2 2 Identified
causes
2 2 2 Table
Fault Tree Analysis (FTA) 2 2 2 2 2 Identified
causes
2 2 2 Graph
Event Tree Analysis (ETA) 2 2 2 2 2 Identified
causes
2 2 2 Tree
Preliminary Risk Analysis (PRA) 2 2 2 2 2 Identified
causes
2 2 2 Table
•
Identify and classify potential and reported ADEs
and MEs (Rq2);
•
Express ADEs in terms of combining potential fac-
tors, situations, events, and consequences (Rq3);
•
Understand the strong relationships between the
ME and activities of the MUP (Rq4);
•
Consider the organizational and human factors
which intervene the ME occurrence (Rq5);
• Produce a dynamic map ranking all ADEs (Rq6);
•
Offer a well designed risk-process knowledge man-
agement repository (Rq7).
•
Provide the EFC team a modeling tool that also
adheres to usability, performance, reliability, scala-
bility, and availability requirements (Rq8).
Consequently, to advance the patient safety, health-
care facilities are required to implement an effective
detection policy, prevention and control of MEs risks
related to the complex MUP. To tackle this situation
we suggest the exploration of new research domains,
in particular the integrated management of risks and
business processes.
4 BPRIM FRAMEWORK
Since several years a major research interest is given
to integrate the two traditionally separated fields of
risk management and business process management
in a common concept known as R-BPM. The impor-
tance of this integration was confirmed by the research
community (Zur Muehlen and Ho, 2005), in industry
guidelines (COSO 2004), and in many literature re-
views (Suriadi et al., 2014; Thabet et al., 2018; Lamine
et al., 2019).
Business Process-Risk Management - Integrated
Method (BPRIM) (Lamine et al., 2019; Thabet et al.,
2018; Sienou, 2009) is one of the important approaches
proposed in the R-BPM context. The method suggests
an integrative approach with three components:
•
A conceptual unification of risk and business pro-
cess based on the coupling between the ISO/DIS
19440 conceptual model and a new conceptual
model for risk.
•
A BPRIM modeling language composed by ab-
stract and concrete syntax. The abstract syntax is
described by a meta-model extending the ISO/DIS
19440 constructs with a new set of constructs for
AdoBPRIM: Towards a New Healthcare Risk-aware Business Process Management Tool
501
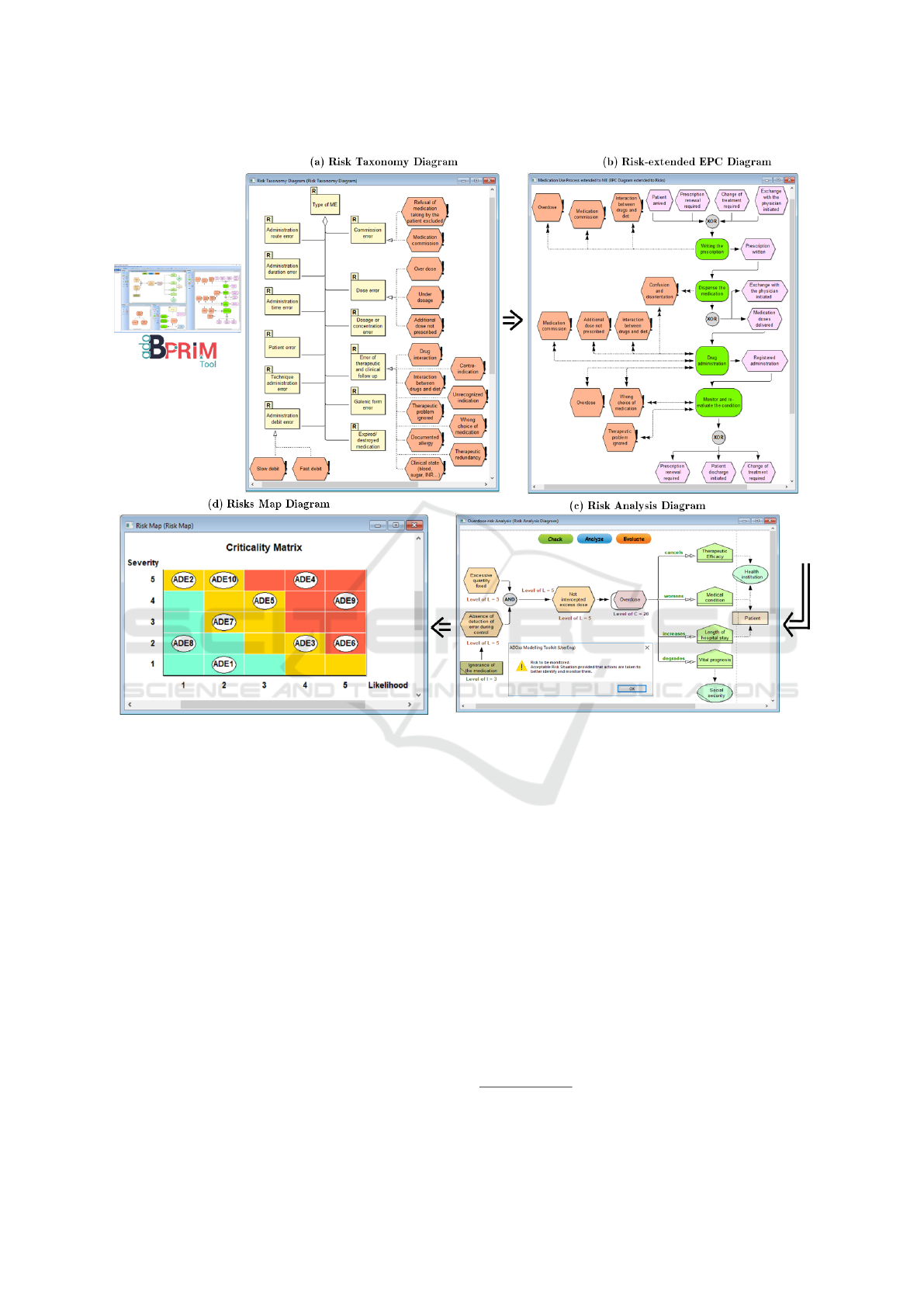
Figure 2: BPRIM Framework (excerpt).
risk modeling. The concrete syntax is proposed by
extending the Event-driven Process Chains (EPC)
notation. Figure 2 shows an extract of the BPRIM
meta-model.
•
A BPRIM lifecycle based on the coupling of the
business process management and the risk man-
agement lifecycles. Considering information ex-
changed between the synchronized lifecycles of
business process design and risk management, a
set of diagrams was identified (e.g. Context, EPC,
Risk-extended EPC, Risk Taxonomy, Risk, Risk
Analysis, Risks Map) (see Figure 2).
In order to evaluate the potential of BPRIM and the
ADOBPRIM tool, we specify for each part individually
to what extent it satisfies the requirements defined in
Section 3.
4.1 ADOBPRIM Tool Development
The modeling tool supporting BPRIM called
ADOBPRIM has been developed as a project within
the Open Models Laboratory (OMiLAB), a worldwide
community of modelers and modeling method devel-
opers (Bork et al., 2019). A free download and further
information on ADOBPRIM are available through the
corresponding project page
1
.
To realize the ADOBPRIM tool, respecting non-
functional requirements (Rq8) previously defined in
Section 3, we have chosen the ADOxx meta-modelling
platform (ADOxx.org, 2019). ADOxx has been
widely used in industry and academia. In the past
twenty years, more than 50 domain-specific model-
ing tools have been successfully realized with ADOxx
(see (Karagiannis et al., 2016) for an overview).
1
ADOBPRIM project page [online]: http://austria.omilab.
org/psm/content/BPRIM/info, last visited: 16.12.2019
HEALTHINF 2020 - 13th International Conference on Health Informatics
502

4.2 ADOBPRIM Models
The main diagrams of our approach are outlined below
and also in Figure 2 b, c, d, and e. Moreover, we
highlight how each ADOBPRIM model contributes to
the identified requirements:
Risk Taxonomy Diagram:
This diagram aims to
build an inventory of potential risks and to classify
them according to various criteria. Risk identifi-
cation can be performed using literature analysis
and/or brainstorming with teamwork.
→ Fulfilling requirement Rq2.
Risks-extended EPC Diagram:
EPC diagrams orig-
inally describe business processes by means of
related and collaborative activities that produce a
specific service or product. Thereby, EPCs provide
a comprehensive understanding of the system and
enable process improvement. The Risks-extended
EPC diagram aims to assign potential risks, de-
fined in the Risk Taxonomy diagram to individual
activities of the EPC diagram.
→
Fulfilling requirements Rq1, Rq4, Rq5, and
Rq7.
Risk Analysis Diagram:
This diagram aims to ana-
lyze and evaluate individual risks. It includes:
(i) Underlying causes and consequences; (ii) Risk
level calculation, and (iii) Risk evaluation. Risk
is a complex concept defined in different ways
depending on the context and the discipline con-
cerned. In our case, a risk R is expressed in terms
of scenario S, likelihood L and severity G as ex-
pressed in Equation 1:
R = (S, L, G) (1)
Risk scenario is expressed in terms of a combi-
nation of potential factors, situations, events, and
consequences. Likelihood level is based on the
expertise, knowledge and actual experience of the
group scoring the likelihood. Severity of impact
indicates the impact of the risk event to values
interested for stakeholders. In this work, we are in-
terested in qualitative risk analysis. A guide to like-
lihood and severity of impact levels is presented
in Table 2. Definitions for the qualitative values
were based on (BLONDEL and BRUNEL, 2004).
The output risk level is illustrated in a two dimen-
sional risk matrix. The purpose of risk evaluation
is to prioritize the risks, and to decide which risks
require treatment as well as the mode of treatment.
→ Fulfilling requirement Rq3.
Risks Map Diagram:
This diagram is a two dimen-
sional risk matrix showing the risk level of each
analyzed risk.
Table 2: Likelihood and severity of impact levels according
to (BLONDEL and BRUNEL, 2004).
Likelihood Severity
1 Very improbable 1 Minor
2 Very unlikely 2 Significant
3 Unlikely 3 Major
4 Possible/Likely 4 Critical
5 Very likely to certain 5 Catastrophic
→ Fulfilling requirement Rq6.
Consequently, through models and capabilities of
the ADOBPRIM tool of the BPRIM method, all func-
tional and non-functional requirements defined in Sec-
tion 3 are satisfied. Next, we will present preliminary
results of the evaluation of the BPRIM framework and
the ADOBPRIM tool in a real case study.
5 PRELIMINARY EXPERIMENT
RESULTS
5.1 Experiment Context
To evaluate capabilities, feasibility, and relevance of
the BPRIM framework and the ADOBPRIM tool, we
use a real-world case study within an existing hospi-
tal. The study was conducted in the Pays d’Autan
Hospital of the InterCommunal Hospital Center of
Castres-Mazamet in France. With more than 388 beds,
this hospital includes 30 wards.
According to the World Health Organization
and (Kane-Gill et al., 2017), MEs and ADEs occur
at a higher frequency and with a greater likelihood for
elderly patients. Improving the quality of medication
use and medication safety for elderly patients is thus
an important public health priority. For this reason,
our case study focuses on ADEs risks management
related to the MUP for elderly patients in the Geriatric
department of the Pays d’Autan Hospital. Our experi-
mentation aims to: (i) check the feasibility of using the
BPRIM framework to manage a serious of ADEs risks
related to the MUP of elderly patients; (ii) experiment
with the ADOBPRIM tool in a real case study; and
(iii) identify potentials for improvement.
The experiment was carried out in collaboration
with several actors of the Pays d’Autan Hospital,
specifically two doctors, three nurses, two pharmacists,
one pharmacy technician, and the Quality Manager.
AdoBPRIM: Towards a New Healthcare Risk-aware Business Process Management Tool
503

5.2 Preliminary Results
The repository of models developed during the experi-
mentation currently holds around 40 BPRIM models.
These last are created and validated in collaboration
with the experimentation team.
The experimentation followed below steps:
1.
Identify organizational units, roles and values re-
lated to the MUP and describe relationships be-
tween them in the context model. As a result, 8
organizational units, 10 roles and 6 values are pre-
sented in the context model;
2.
Build AS-IS models of the MUP of elderly pa-
tients in the Geriatric department: First a value-
added chain model describing an overview of the
MUP sub-processes is presented, and then each
sub-process is defined by an EPC model. As a
result, 14 EPC models presenting a detailed func-
tional and organizational view of the MUP in the
Geriatric department are presented. The EPC mod-
els involve up to 46 activities.
3.
Identify potential and reported ADEs in the depart-
ment. As a result, 16 ADEs organized in 12 classes
are presented in the Risk Taxonomy model (see
Figure 2 a);
4.
Extend AS-IS models with potential and reported
ADEs in the department. As a result, 14 EPC
models extended with the 16 potential ADEs pre-
viously defined. Figure 2 b) shows an example of
an Risk-extend EPC model;
5.
For each ADE, analyze potential and reported
facts, risk situations and risk events contributing
to the ADE occurrence. We studied 10 serious
ADEs risks, reported by professionals, related to
the MUP of elderly patients and that had analyzed
by the EFC using the ALARM method. As a result,
10 Risk Analysis models and 10 Risk models cor-
responding to reported ADEs are defined. Figure 2
c) shows the Overdose Risk Analysis model;
6.
Evaluate each ADE using the risk matrix accep-
tance defined by (BLONDEL and BRUNEL, 2004)
and produce a Risks Map model ranking all ADEs
by risk level (see Figure 2 d);
7.
Diagnose obtained results looking for potential
improvements of the AS-IS models.
ADOBPRIM is a reactive/proactive integrated man-
agement tool of risk and business process management
dedicated to facilitate and to support the multidisci-
plinary EFC members in their ADEs analysis exercise.
Indeed, the tool supports them in understanding the
MUP activities, identifying ADEs, understanding how
the ADE has occurred, prioritizing remedial interven-
tions, and possibly enhancing the MUP steps.
Moreover, with the ADOBPRIM tool, a database
of potential ADEs, analyzes already carried out during
previous EFCs can be accessed and augmented with
new data. In addition, the tool allows accelerating
the ADEs analysis process which consequently allows
analysing more reported ADEs per EFC meeting. In-
deed, due to the heaviness and the complexity of risk
management methods currently used in healthcare fa-
cilities, the EFC committee chooses only one ADE per
meeting in order to analyze it thoroughly.
The tool usage guidelines are partly defined by the
BPRIM lifecycle. It should also be noted that a training
session on the tool usage is necessary for a first use.
Some models obtained by ADOBPRIM are presented
like in figure 2. The latter effectively illustrates some
of the models applied to the MUP extended to the risks
of occurrence of MEs.
Validation and evaluation of our implementation
results are currently performed in accordance with
the very involved healthcare professionals of the Pays
d’Autan Hospital.
6 CONCLUSIONS
The main contribution of this paper is the implementa-
tion of an effective approach to manage MEs related to
the Medication Use Process based on the R-BPM con-
cept. We presented an R-BPM framework for risk and
business process management comprising the BPRIM
method and the ADOBPRIM modeling tool.
The validity check for our contribution was per-
formed in a real case study. The results motivate us to
pursue the improvement of this method by integrating
additional mechanisms and algorithms of risks analysis
of the ADE related to the Medication Use Process of a
patient within the healthcare facilities and studying the
potential of the application of our framework in other
healthcare processes as part of our future research.
ACKNOWLEDGEMENTS
The authors would like to thank the experimentation
team from the Pays d’Autan Hospital of the Intercom-
munal Hospital Center (CHIC) of Castres-Mazamet
and more specifically Dr. CUFI, the head of the Geri-
atric Department and Dr. CLEOSTRATE, the head
of Inpatient Pharmacies who allowed us to evaluate
our BPRIM method and ADOBPRIM tool on a real
case and provided insights and expertise that greatly
enhanced this research. We would also like to thank
HEALTHINF 2020 - 13th International Conference on Health Informatics
504

the OMiLAB team for assistance with the ADOxx plat-
form and for providing a collaborative space on their
OMiLAB portal.
REFERENCES
ADOxx.org (2019). The adoxx metamodeling platform.
www.adoxx.org, last accessed: 17.12.2019.
Barker, K. N., Flynn, E. A., Pepper, G. A., Bates, D. W., and
Mikeal, R. L. (2002). Medication errors observed in
36 health care facilities. Archives of internal medicine,
162(16):1897–1903.
Bates, D. W. (2007). Preventing medication errors: a sum-
mary. American Journal of Health-System Pharmacy,
64(14 Supplement 9):S3–S9.
Bates, D. W., Boyle, D. L., Vander Vliet, M. B., Schneider,
J., and Leape, L. (1995). Relationship between medica-
tion errors and adverse drug events. Journal of general
internal medicine, 10(4):199–205.
BLONDEL, P. and BRUNEL, C. (2004). Etude et
hi
´
erarchisation du risque infectieux nosocomial selon
la criticit
´
e: nouvelle approche. Risques & qualit
´
e en
milieu de soins, (3):17–23.
Bork, D., Buchmann, R. A., Karagiannis, D., Lee, M., and
Miron, E.-T. (2019). An Open Platform for Model-
ing Method Conceptualization: The OMiLAB Digital
Ecosystem. Communications of the Association for
Information Systems, 44:pp. 673––697.
Caporossi, A., Brudieu, E., Lehmann, A., Seigneurin, A.,
and Fran
c¸
ois, P. (2014). An experience feedback com-
mittee for improving medication process safety: an
observational study in a hospital pharmacy department.
J Pharma Care Health Sys S1-010. doi, 10:2.
Committee on Quality of Health Care in America and Insti-
tute of Medicine (2000). To err is human: Building a
safer health system. National Academies Press Wash-
ington, DC.
Committee on Quality of Health Care in America and Insti-
tute of Medicine (2006). Preventing medication errors:
quality chasm series.
Committee on Quality of Health Care in America and In-
stitute of Medicine Staff (2001). Crossing the quality
chasm: A new health system for the 21st century. Na-
tional Academies Press Washington, DC.
Francois, P., Sellier, E., Imburchia, F., and Mallaret, M.
(2013). Experience feedback committee: a method for
patient safety improvement. Revue d’epidemiologie et
de sante publique, 61(2):155–161.
HAS, H. A. d. S. (2013). Outils de s
´
ecurisation et d’auto-
´
evaluation de l’administration des m
´
edicaments. Saint-
Denis La Plaine: HAS.
Kane-Gill, S. L., Dasta, J. F., Buckley, M. S., Devabhakthuni,
S., Liu, M., Cohen, H., George, E. L., Pohlman, A. S.,
Agarwal, S., Henneman, E. A., et al. (2017). Clini-
cal practice guideline: safe medication use in the icu.
Critical care medicine, 45(9):e877–e915.
Karagiannis, D., Mayr, H. C., and Mylopoulos, J., editors
(2016). Domain-Specific Conceptual Modeling, Con-
cepts, Methods and Tools. Springer.
Lamine, E., Thabet, R., Seinou, A., Bork, D., Fontanili,
F., and Pingaud, H. (2019). Bprim: An integrated
framework for business process management and risk
management. Computers in Industry, page in press.
Leape, L. L., Brennan, T. A., Laird, N., Lawthers, A. G.,
Localio, A. R., Barnes, B. A., Hebert, L., Newhouse,
J. P., Weiler, P. C., and Hiatt, H. (1991). The nature
of adverse events in hospitalized patients: results of
the harvard medical practice study ii. New England
journal of medicine, 324(6):377–384.
Lisby, M., Nielsen, L. P., Brock, B., and Mainz, J. (2010).
How are medication errors defined? a systematic liter-
ature review of definitions and characteristics. Interna-
tional Journal for Quality in Health Care, 22(6):507–
518.
Moyen, E., Camir
´
e, E., and Stelfox, H. T. (2008). Clinical
review: medication errors in critical care. Critical
Care, 12(2):208.
NCC MERP, N. C. C. f. M. E. R. and Prevention (2019).
What is a medication error?
Sienou, A. (2009). Proposition d’un cadre m
´
ethodologique
pour le management int
´
egr
´
e des risques et des proces-
sus d’entreprise. PhD thesis, Institut National Poly-
technique de Toulouse.
Suriadi, S., Weiß, B., Winkelmann, A., ter Hofstede, A.
H. M., Adams, M., Conforti, R., Fidge, C., Rosa, M. L.,
Ouyang, C., Rosemann, M., Pika, A., and Wynn, M.
(2014). Current Research in Risk-Aware Business Pro-
cess Management - Overview, Comparison and Gap
Analysis. Communications of the AIS (CAIS), 34(Jan-
uary 2014).
Thabet, R., Lamine, E., Boufaied, A., Korbaa, O., and Pin-
gaud, H. (2018). Towards a risk-aware business process
modelling tool using the adoxx platform. In Interna-
tional Conference on Advanced Information Systems
Engineering, pages 235–248. Springer.
Vest, T. A., Gazda, N. P., Schenkat, D. H., and Eckel, S. F.
(2019). Practice-enhancing publications about the med-
ication use process in 2017. American Journal of
Health-System Pharmacy.
Zur Muehlen, M. and Ho, D. T.-Y. (2005). Risk manage-
ment in the bpm lifecycle. In International Confer-
ence on Business Process Management, pages 454–
466. Springer.
AdoBPRIM: Towards a New Healthcare Risk-aware Business Process Management Tool
505
