
Optical Technology for Ultraviolet Erythema Assessment and
Minimal Erythema Dose Determination in Healthy Volunteers
Mikhail Makmatov-Rys
1
, Alexey Glazkov
1
, Irina Raznitsyna
1
, Dmitriy Kulikov
1,3
,
Anton Molochkov
1
, Albina Khlebnikova
1
, Ekaterina Kaznacheeva
2
, Alexey Sekirin
1
and Dmitry Rogatkin
1
1
Moscow Regional Research and Clinical Institute "MONIKI", 61/2,
Shchepkina Str., Moscow, RF, 129110, Russian Federation
2
Cosmetological Clinic «Lemark», Voronezh, 32 Vladimir Nevsky Str., RF, 394088, Russian Federation
3
Institute of Public Health named after N.A. Semashko,
12/1c1 Vorontsovo Pole Str., Moscow, RF, 105064, Russian Federation
Keywords: Ultraviolet Erythema, Minimal Erythema Dose, Inflammation, Fluorescence, Saturation, Non-invasive,
Diagnostics.
Abstract: Currently, in clinical practice, the assessment of ultraviolet (UV) -induced erythema and the determination
of the minimal erythema dose (MED) is done visually, which is subjective, inaccurate and associated with
high variability of the results. To solve this problem, the application of optical methods seems promising,
allowing us to evaluate changes in epidermis and dermis induced by UV exposure. In this study the analysis
of endogenous fluorescence and microcirculation characteristics by non-invasive optical methods revealed
the relationship between the intensity of endogenous fluorescence of porphyrins and oxygen consumption
with a dose of UV radiation. The correlation of the intensity of endogenous fluorescence of the irradiated
region normalized to intact tissue with a dose of UV was demonstrated. Therefore, optical diagnostic methods
can be a promising tool for non-invasive and quantitative assessment of UV erythema and MED.
1 INTRODUCTION
The assessment of skin reaction on different doses of
ultraviolet radiation (UVR) with different
wavelengths is one of the challenging issues of
modern photobiology and medicine. Currently, the
traditional method of assessing the degree of
exposure to UV radiation in humans and animals is
based on the calculation of the minimal erythema
dose. Minimal erythema dose (MED) is an amount of
UV exposure leading to the development of
minimally perceptible erythema on untanned skin
within 24 hours after irradiation (Heckman et al.,
2013). Thus, determination of MED is based on
assessment the charactheristics of the
pathophysiologic phenomenon - UV-erthema
(Makmatov-Rys et al., 2019). MED is usually
measured in mJ/cm
2
. MED is widely used in clinical
practice and experiments in the evaluation of
photosensitivity. It is applied to determine the UVA
and UVB starting doses in the phototherapy of skin
diseases (Krutmann et al., 2008). Moreover, MED
assessment is one of the diagnostic methods for some
photodermatoses (Hönigsmann, 2008).
MED is traditionally determined visually by
naked eye, which is a subjective and inaccurate
method (Lock‐Andersen et al., 1996). For instance,
Falk M. and Ilias M. showed that the agreement
between observers on the characteristics of UV
erythema, was excellent for skin redness with a sharp
border, but for reactions with a diffuse or indistinct
border there was a substantial inter-observer
variability. Mistakes can occur during the visual
assessment of the Fitzpatrick skin type especially in
tanned patients or in dark skin when the evaluation is
made by untrained doctor (Falk et al., 2008).
Incorrect determination of MED can lead to an
overestimation of the dose of UV radiation in the
starting point of phototherapy course and to such
complications as burns, hyperpigmentation, dry skin,
herpes simplex reactivation, and in some cases to
aggravation of the underlying skin disease. In
addition, the occurrence of such complications leads
to an interruption in the phototherapy course, an
Makmatov-Rys, M., Glazkov, A., Raznitsyna, I., Kulikov, D., Molochkov, A., Khlebnikova, A., Kaznacheeva, E., Sekirin, A. and Rogatkin, D.
Optical Technology for Ultraviolet Erythema Assessment and Minimal Erythema Dose Determination in Healthy Volunteers.
DOI: 10.5220/0009177000730078
In Proceedings of the 13th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2020) - Volume 1: BIODEVICES, pages 73-78
ISBN: 978-989-758-398-8; ISSN: 2184-4305
Copyright
c
2022 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
73

increase in the numer of clinic visits and the
associated economic costs. In the long term, the risk
of malignant skin neoplasms and photoaging
increases.
To avoid the limitations of conventional methods
for MED assessment additional instrumental
methods, in particular optical, are used.
There are data in the literature on the use of the
following optical methods for assessing erythema
intensity and MED: reflective spectroscopy
(Bodekær et al., 2013), colorimetry (Jeon et al.,
2014), laser doppler flowmetry (Falk et al. 2008),
laser doppler visualization (Wilhelm et al., 2001), and
confocal microscopy (Yamashita et al., 2007).
Despite the progress in the field of the objective
assessment of UV erythema and MED, the
abovementioned methods have some limitations.
Current studies are trying to find a correlation
between objective optical metrics and subjective
visual characteristics of erythema, such as color,
distinctness of borders, contours. Unfortunately, the
examination of pathophysiological mechanisms of
severe UV-induced tissue damage is beyond the
scope of these studies.
Meanwhile, laser fluorescence spectroscopy
(LFS) has potential applications in this field.
Fluorophores responsible for inflammation and
hypoxia which play a role in UV-induced skin
damage, could be detected by LFS in red and green
spectrum range (Franco et al., 2016). The literature
describes the use of laser fluorescence spectroscopy
in vivo in experimental models for assessment local
inflammation (Petritskaya et al., 2015), radiation skin
damage (Raznitsyna et al., 2018) and the skin fibrosis
(Chursinova et al., 2019). There are studies on the use
of fluorescence spectroscopy to assess structural skin
changes during chronic UV damage (Tian et al.,
2001). Papazoglou E. et al. (2010) compared the data
of LFS, skin morphology and expression of matrix
metalloproteinase 13 (MMP-13) to assess changes
caused by prolonged exposure to UVB radiation on
the skin of hairless mice. The authors found that LFS
can be used to estimate epidermal thickness and
fluorescence parameters correlates with tryptophan
expression and cell proliferation and may indicate the
presence of “burn cells” in the epidermis (Papazoglou
et al., 2010)
The aim of this study was to assess the
applicability of complex optical technologies
including LFS and optical tissue oximetry in the
assessment of ultraviolet-induced skin damage and
MED at different time periods after UV irradiation
2 MATERIALS AND METHODS
The study was conducted on a group of healthy
volunteers (n = 14, 8 male and 6 female) aged 26 ± 3
years with Fitzpatrick skin phototypes II and III. In
all participants traditional MED assessment method
(described by Heckman et al. (2013)) was perfomed
on the skin of the upper back or on the skin of the
abdomen. UVB irradiation was performed using a Dr.
Honle Dermalight 500-1 series (manufactured by Dr.
Honle Medical Technology GmbH, Germany),
equipped with Phillips UV-B Narrowband PL lamps
with a wavelength of 311 nm. The Daavlin DosePatch
hypoallergenic plate with six square windows (a
square size of 1ⅹ1 cm
2
) was attached to the skin of the
back or abdomen, the distance to the UV source was
30 cm. The UV intensity was measured using a
Waldmann Variocontrol spectroradiometer (UV
meter). The dose of UV radiation from cell to cell
increased stepwise depending on the phototype of the
skin of the subject according to reference tables
(Palmer et al., 2005). The skin in the windows was
cumulatively exposed to UV radiation in the range
from 100 to 770 mJ/cm
2
. 24 hours after UV-B
exposure, the participants in the experiment were
conducted a subjective visual assessment of erythema
by 2 observers. The erythema reaction was graded
using a visual rating scale (Faurschou, et al., 2009).
Based on the results of the visual examination, the site
corresponding to the MED (barely noticeable
erythema) was determined and the dose of UVB was
calculated. Detailed characteristics of MED and
phototype participants are presented in table 1.
Before UVB irradiation and after 0.5, 3, 6, 24
hours after it, on the skin in each of 6 square windows
and on the contralateral area of intact skin (1 cm2),
the endogenous fluorescence of porphyrins was
evaluated by LFS and local blood flow characteristics
was measured by optical tissue oximetry (OTO)
implemented in the LAKK-M system (SPE 'LAZMA'
Ltd, Russia), as described in (Chursinova et al.,
2019).
The diagnostic system scheme is shown in Figure
2. The choice of the abovementioned time points was
based on an analysis of the literature on the
pathogenesis of the of acute UV damage (Hruza et al.,
1993). The process of measuring of optical
parameters is presented in Figure 1.
BIODEVICES 2020 - 13th International Conference on Biomedical Electronics and Devices
74

Figure 1: Process of optical measurements of the abdomen
skin after irradiation with ultraviolet B.
Figure 2: Scheme of the diagnostic system.
As a rule, the maximum absorption of most
endogenous fluorophores is observed in the UV
wavelength range. However, the radiation of this
range has a low penetrating power (less then 0.1 mm
(Mustafa et al., 2013) as distinct from the visible part
of the spectrum. Therefore, the spectra of secondary
radiation (backscattered and fluorescence) were
recorded from each region of interest after its
irradiation by low-power laser sources with
wavelengths λ
e
= 635 nm and λ
e
= 535 nm.
Porphyrin is characterized by a two-hump
fluorescence spectrum with maxima at wavelengths
of 625–630 and 700-710 nm (Croce et al., 2014). In
the wavelength range of 650 - 750 nm, porphyrins
make the main contribution to the endogenous
fluorescence of biological tissue, but at a wavelength
of 625–630 nm fluorescence of porphyrins is more
pronounced.
The fluorescence intensities I
f
of porphyrins were
estimated at wavelengths λ
f
= 710 to verify theirs
presence and at λ
f
= 630 nm to quantitative
assessment, respectively. Despite the fact that other
fluorophores (for example, lipofuscin) can also
fluoresce in the range of 625–630 nm, their
contribution to the total intensity insignificant.
To exclude the variability of the initial
endogenous fluorescence of volunteers’ skin, the
fluorescence intensity was normalized to the intact
region μ(λ
f
):
μ(λ
f
) = I(λ
f
)/ I
0
(λ
f
) (1)
where I(λ
f
) is the fluorescence intensity from the
iradiated area, I
0
(λ
f
) is the fluorescence intensity from
the intact area.
To evaluate the parameters of local blood flow,
blood filling volume (V
b
) and tissue oxyhemoglobin
saturation (S
t
O
2
) were recorded for each region of
interest for 20 seconds. Then, according to the time-
averaged data the specific oxygen consumption of the
tissues U characterized by the oxygen intake per
tissue blood flow volume unit was calculated with the
use of the following formula (Rogatkin et al., 2013):
U= (S
p
O
2
- S
t
O
2
)/ V
b
(2)
In this formula S
p
O
2
is the functional pulse
saturation of the oxyhaemoglobin fraction in the
arterial peripheral blood. It was assumed equal to
98%.
In the intact skin area, a melanin index (MI) was
measured for each participant using a
spectrophotometric instrument. «Spectrotest» (SPE
‘Cyclone-Test’ Ltd, Russia) (Afanasyev et al., 2007).
Results of measurements are showed in Table 1.
Table 1: Characteristics of volunteers enrolled in the study.
N Phototype MI
MED,
mJ/cm2
Site of
MED
assessment
1 3 0.0524 280 abdomen
2 3 0.0566 400 upper bac
k
3 2 0.0445 280 upper bac
k
4 2 0.0501 200 abdomen
5 3 - 470 abdomen
6 2 0.0501 280 u
pp
er bac
k
7 3 0.0560 750 u
pp
er bac
k
8 2 0.0544 560 upper bac
k
9 3 0.0574 380 upper bac
k
10 3 0.0693 750 u
pp
er bac
k
11 3 0.0693 770 u
pp
er bac
k
12 2 - 280 u
pp
er bac
k
13 3 - 380 upper bac
k
14 3 - 380 abdomen
Statistical analysis was performed in Microsoft
Excel 2016 and Statistica 12 (Statsoft inc., USA). The
analysis of dynamic changes in the optical parameters
described above was carried out using the Wilcoxon
Optical Technology for Ultraviolet Erythema Assessment and Minimal Erythema Dose Determination in Healthy Volunteers
75
test. The relationship between the obtained optical
data and the dose of UV radiation was evaluated using
the Spearman rank correlation coefficient. The
probability of an error of the first kind was considered
statistically significant to be less than 5% (p <0.05).
3 RESULTS AND DISCUSSION
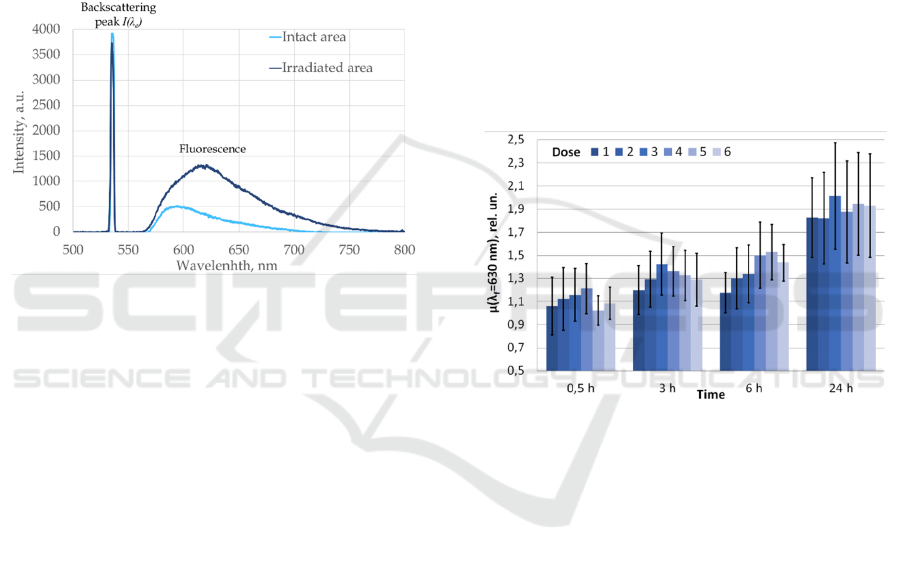
Examples of measured fluorescence spectra from the
intact (non-irradiated) and irradiated skin sites at λ
e
=
630 nm after 24 hours after the UV-irradiation is
shown in the Figure 3.
Figure 3: The example of the fluorescence spectra in intact
and irradiated skin after 24 hours after UVB exposure; λe =
630 nm.
Using Spearman's rank correlation coefficient,
positive correlation relationships were revealed
between the cumulative dose of UV and the specific
oxygen consumption of the tissues (U) normalized to
intact skin after 3 hours (correlation coefficient [r] =
0.297; p = 0.01) and 24 hours (r = 0.307; p = 0.0004)
after the irradiation. In addition, a positive correlation
was found between the total UV dose and the
fluorescence intensity of porphyrins λ
e
= 630 nm 6
hours after UV irradiation normalized to intact skin (r
= 0.249, p = 0.01).
The findings may reflect the course of acute UV-
induced skin damage. Thus, the U index reflects an
increase in the metabolic activity of skin cells
susceptible to acute UV damage. Under the influence
of UV, mast cell degranulation, vascular endothelial
damage, vasoditalation are observed, vasoactive
substances are released - histamine, nitric oxide,
arachidonic acid derivatives, which also contribute to
the formation of infiltrate from immune cells in the
affected area (Clydesdale et al., 2001). Logan and
colleagues showed that one consequence of UV
exposure of the skin is damage to epidermal cells
which becomes evident as early as 2 hours after UV
irradiation (Logan & Wilhelm, 1963). One study
showed that peak infiltration of leucocytes after UVB
irradiation occurs at 4-6 hours and the response
concludes after 48 hours (Logan & Wilhelm, 1966).
It has also been shown that with increasing intensity
and dose of UV radiation, skin damage becomes more
pronounced (Hruza, 1993)
In addition, according to the results of the study,
it was found that normalized fluorescence intensity
and tissue content index in all irradiated skin sites
regularly changed stepwise over time. The most
significant increase in the intensity of fluorescence of
porphyrins in green light (λ
e
= 630 nm), normalized
to intact skin in all 6 cells was observed 24 hours
compared with 0,5 hours after UV exposure (Figure
4).
These results allow us to hypothesize that UV
exposure affects the metabolism and accumulation of
porphyrins in the skin.
Figure 4: The dynamics of the porphyrin's intensity
normalized to intact tissue in skin sites irradiated with
stepwise increasing doses of UVB (6 – lowest dose, 1 –
highest dose) in 4 time points after UV-exposure; λe = 630
nm.
Additionally, using Spearman's rank correlation
coefficient, it was found that the MED of volunteers
correlated with Fitzpatrick's skin phototype (r = 0.56;
p= 0.036), the melanin index (MI) showed correlation
with the skin phototype (r = 0.79; p= 0.007). This
results corresponds with previously to published data:
for instance, D.L. Damian and colleges in their study
showed good correlation between MI, Fitzpatrick
phototype and MED in 60 healthy volunteers
(Damian et al 1997).
It is important to mention that our work have some
limitations. We didn’t include subjects with darker
skin phototypes (IV-VI) in study population. It is
known that melanin content is significantly higher in
the skin of subjects with darker skin type (Lu et al.,
1996).
BIODEVICES 2020 - 13th International Conference on Biomedical Electronics and Devices
76

Melanin is known to absorb the radiation of the
visible spectrum, which reduces the registered signal
significantly. In these cases increasing the power of
the laser radiation may increase the signal-to-noise
ratio and solve the problem. But in further studies it
is important to estimate the minimum laser power for
skin phototypes IV-VI at which peaks of endogenous
fluorophores can be distinguished.
Also, since this study involved young patients of
approximately the same age, it is also necessary to
conduct the similar studies with subjects of different
age groups. Lipofuscin age pigments accumulate in
cells with age, also has fluorescent properties (Brunk
et al., 2002). Therefore, it is necessary to evaluate its
contribution to the total skin spectrum and the
possibility of reliable identification of porphyrins in
older people.
4 CONCLUSIONS
The results of this pilot study showed that the
integrated application of the LFS and OTO methods
for objective non-invasive assessment of erythema
has prospects for further investigation in larger
studies. This techniques give us opportunity to access
pathophisiological alternations (e.g. inflmmation and
vasodilatation) taking place in the skin after acute UV
damage. To gain more precise data, it is worth to
analyze optical parameters of of the skin of different
anatomical zones irradiated with UV (for example,
back and abdomen) in a larger groups of young
volunteers darker skin phototypes.
In the future, these developments may become the
basis for the development of diagnostic systems for
quantitative predictive assessment of MED.
ACKNOWLEDGEMENTS
The reported study was funded by RFBR, project
number 20-32-70134
REFERENCES
Afanasyev, A.I., Rogatkin, D.A., Sergienko, A.A., &
Shumsky, V.I. 2007 A new generation of non-invasive
spectrophotometry instruments: an optical tissue
oximeter and an analyzer of volumetric blood filling of
soft biological tissues “Spectrotest”. Doctor. ru, (4), 45-
47. (In Russian)
Bodekær, M., Philipsen, P. A., Karlsmark, T., & Wulf, H.
C. 2013. Good agreement between minimal erythema
dose test reactions and objective measurements: an in
vivo study of human skin. Photodermatology,
photoimmunology & photomedicine, 29(4), 190-195.
Brunk, U. T., & Terman, A. (2002). Lipofuscin:
mechanisms of age-related accumulation and influence
on cell function. Free Radical Biology and Medicine,
33(5), 611-619.
Chursinova Y.V., Kulikov D.A., Rogatkin D.A.,
Raznitsyna I.A., Mosalskaya D.V., Bobrov M.A.,
Petritskaya E.N., Molochkov A.V., 2019. Laser
fluorescence spectroscopy and optical tissue oximetry
in the diagnosis of skin fibrosis. Biomedical Photonics,
8(1), 38-45.
Clydesdale, G. J., Dandie, G. W., & Muller, H. K. (2001).
Ultraviolet light induced injury: immunological and
inflammatory effects. Immunology and cell biology,
79(6), 547-568.
Croce A. C., & Bottiroli, G., 2014. Autofluorescence
spectroscopy and imaging: a tool for biomedical
research and diagnosis. Eur J Histochem, 58(4): 2461,
320-337.
Damian, D. L., Halliday, G. M., & Barnetson, R. S. (1997).
Prediction of minimal erythema dose with a reflectance
melanin meter. British Journal of Dermatology, 136(5),
714-718.
Falk, M., Ilias, M., & Anderson, C. 2008. Inter‐observer
variability in reading of phototest reactions with sharply
or diffusely delineated borders. Skin Research and
Technology, 4(4), 397-402
Faurschou, A., Wulf, H. C., 2009. European Dermatology
Guideline for the photodermatoses. 2.
Phototesting.EDF guidelines for dermatology in
Europe. Berlin: ABW Wissenschaftsverlag.
Franco, W., Gutierrez-Herrera, E., Kollias, N., Doukas, A.,
2016. Review of applications of fluorescence excitation
to spectroscopy dermatology. British Journal of
Dermatology, 174 (3), 499-504
Heckman, C. J., Chandler, R., Kloss, J. D., Benson, A.,
Rooney, D., Munshi, T., Oslin, D. W. 2013). Minimal
erythema dose (MED) testing. JoVE (Journal of
Visualized Experiments), (75), e50175.
Hönigsmann, H. 2008 Polymorphous light eruption.
Photodermatology, photoimmunology &
photomedicine, 24(3), 155-161.
Hruza, L. L., & Pentland, A. P. 1993. Mechanisms of UV-
induced inflammation. Journal of Investigative
Dermatology, 100(1), 35-41.
Jeon, S. Y., Lee, C. Y., Song, K. H., Kim, K. H. 2014.
Spectrophotometric measurement of minimal erythema
dose sites after narrowband ultraviolet B phototesting:
clinical implication of spetrophotometric values in
phototherapy. Annals of dermatology, 26(1), 17-25.
Krutmann, J., & Hönigsmann, H. 2009 Dermatological
phototherapy and photodiagnostic methods. C. A.
Elmets, & P. R. Bergstresser (Eds.). Berlin:: Springer.
Lock‐Andersen, J., & Wulf, H. C. 1996. Threshold level for
measurement of UV sensitivity: reproducibility of
phototest. Photodermatology, photoimmunology &
photomedicine, 12(4), 154-161.
Optical Technology for Ultraviolet Erythema Assessment and Minimal Erythema Dose Determination in Healthy Volunteers
77

Logan, G., & Wilhelm, D. L. 1963. Ultraviolet injury as an
experimental model of the inflammatory reaction.
Nature, 198(4884), 968.
Logan, G., & Wilhelm, D. L. 1966. Vascular permeability
changes in inflammation. I. The role of endogenous
permeability factors in ultraviolet injury. British
journal of experimental pathology, 47(3), 300.
Lu, H., Edwards, C., Gaskell, S., Pearse, A., & Marks, R.
1996. Melanin content and distribution in the surface
corneocyte with skin phototypes. British Journal of
Dermatology, 135(2), 263-267.
Makmatov-Rys M., Kulikov D., Kaznacheeva E., &
Khlebnikova, A., 2019. Features of the pathogenesis of
acute ultraviolet-induced skin damage, Klinicheskaya
Dermatologia I Venerologia 18(4).
Mustafa, F. H., Jaafar, M. S. 2013. Comparison of
wavelength-dependent penetration depths of lasers in
different types of skin in photodynamic therapy. Indian
Journal of Physics,87(3), 203-209.
Palmer R., Garibaldinos T., Hawk J. 2005 Phototherapy
guidelines. St John’s Institute of Dermatology/ St
Thomas’ Hospital, London
Papazoglou, E., Sunkari, C., Neidrauer, M., Klement, J. F.,
& Uitto, J. 2010. Noninvasive Assessment of UV‐
induced Skin Damage: Comparison of Optical
Measurements to Histology and MMP Expression.
Photochemistry and photobiology, 86(1), 138-145.
Petritskaya, E.N., Kulikov, D.A., Rogatkin, D.A., Guseva,
I.A., and Kulikova, P.A., 2015. Use of fluorescence
spectroscopy for diagnosis of hypoxia and
inflammatory processes in tissue. Journal of Optical
Technology, 82 (12), 810-814.
Raznitsyna, I., Kulikova, P., Rogatkin, D., Kulikov, D.,
Bychenkov, O., Chursinova, Y., Bobrov, M., Glazkov,
A. 2018. Fluorescence of radiation-induced tissue
damage. International journal of radiation
biology, 94(2), 166-173.
Rogatkin, D., Shumskiy, V., Tereshenko, S., Polyakov, P.,
2013. Laser-based non-invasive spectrophotometry–An
overview of possible medical applications. Photonics &
Lasers in Medicine, 2(3), 225-240.
Tian, W. D., Gillies, R., Brancaleon, L., & Kollias, N. 2001.
Aging and effects of ultraviolet A exposure may be
quantified by fluorescence excitation spectroscopy in
vivo. Journal of investigative dermatology, 116(6),
840-845.
Wilhelm, K. P., Kaspar, K., & Funkel, O. 2001.
Comparison of three techniques for evaluating skin
erythemal response for determination of sun protection
factors of sunscreens: high resolution laser Doppler
imaging, colorimetry and visual scoring.
Photodermatology, photoimmunology &
photomedicine, 17(2), 60-65.
Yamashita, T., Akita, H., Astner, S., Miyakawa, M., Lerner,
E. A., & González, S. 2007. In vivo assessment of
pigmentary and vascular compartments changes in
UVA exposed skin by reflectance‐mode confocal
microscopy. Experimental dermatology, 16(11), 905-
911.
BIODEVICES 2020 - 13th International Conference on Biomedical Electronics and Devices
78
