
Design of an Innovative Medical Device to Improve Quality of Life in
Lymphedema Patients
Katherine Wang
1a
and Angeliki Kopsini
2b
1
Department of Mechanical Engineering, University College London, Torrington Place, London WC1E 7JE, U.K.
2
Medical and Scientific Affairs and Healthcare Development Department, Roche Diagnostics Hellas, Athens, Greece
Keywords: Lymphedema, Medical Device, Breast Cancer, Rehabilitation.
Abstract: Lymphedema is a disease that is a common side effect of breast cancer, affecting up to 70% of breast cancer
patients. Currently there is no known curative treatment; management of the disease is based on symptomatic
therapy such as Complete Decongestive Therapy (CDT). Current devices and compression garments used in
CDT are often heavy and cause discomfort and bacterial infections due to poor skin care. Along with the long-
term physical problems, it also causes serious psychological and social problems for patients, affecting their
quality of life significantly. There is an urgent need for innovative approaches for the treatment of lymphedema.
In this paper we propose a novel solution in the form of a light portable pneumatic device “Lymphmotion”, along
with a complimentary compression garment designed to reduce bacterial infection to address this problem.
1 INTRODUCTION
Lymphedema is a disease caused by the inability of
the lymphatic system to transport lymph fluid out of
the affected area, causing accumulation of fluid in
interstitial tissues that results in swelling of the
affected area (Taghian et al., 2014). Breast Cancer
Related Lymphedema (BCRL) is a common side
effect of breast cancer treatment, affecting up to 70%
of breast cancer patients. The number of individuals
coping with this disease continues to increase,
estimated to affect more than 120 million individuals
worldwide (International Lymphedema Framework,
ILF).
It is a leading cause of disability worldwide, yet it
remains underdiagnosed, under-researched, and
underfunded in most health systems (Douglass and
Kelly-Hope, 2019; Keast et al., 2015). The disparity
between diagnosis, treatment and funding could be
due to a lack of public awareness of the condition,
insufficient education and knowledge on
management, and limited financial coverage to
support appropriate methods and materials. It is
suggested that lymphedema may be a common and
under-reported morbidity.
a
https://orcid.org/0000-0002-6900-3213
b
htps://orcid.org/0000-0003-2310-2117
Lymphedema patients have chronic, progressive
swelling, recurrent infections, and significantly
decreased quality of life that affects both physical and
psychological aspects of a patient’s life. The
progressive nature means that the swelling can lead to
disfigurement, disability, decreased limb strength,
mobility, pain, heaviness, and in some cases even
death (Cemal et al., 2011; Keast et al., 2015; Taghian
et al., 2014). Anxiety and frustration are well
documented in patients with secondary lymphedema
(Hayes et al., 2009). Reduced productivity of the
patient, and higher cost of treatment is also a
significant socioeconomic burden; treatment cost of
breast cancer survivors with lymphedema
demonstrated a $10,000 increase in annual treatment
costs compared breast cancer survivors without
(Brahma and Yamamoto, 2019; Cemal et al., 2011;
Shih et al., 2009).
Treatment of lymphedema is largely palliative,
with no cure. The gold standard is Complete
Decongestive Therapy (CDT), a treatment programme
that includes pneumatic devices. Currently there is no
portable pneumatic device for the treatment of
lymphedema, and both researchers and physicians
agree that there is an urgent
need for innovative
Wang, K. and Kopsini, A.
Design of an Innovative Medical Device to Improve Quality of Life in Lymphedema Patients.
DOI: 10.5220/0009369703230328
In Proceedings of the 13th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2020) - Volume 1: BIODEVICES, pages 323-328
ISBN: 978-989-758-398-8; ISSN: 2184-4305
Copyright
c
2022 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved
323

approaches in the treatment of lymphedema.
In this paper, we will give a brief review into the
current state of the art treatment and market overview
for lymphedema, and the patient perspective. We will
also introduce a solution to the current issues with
treatment patients face, in the form of a lightweight
portable pneumatic compression device and garment
named ‘Lymphmotion’.
2 STATE OF THE
ARTTREATMENT AND
PATIENT PERSPECTIVES
2.1 State of the Art
Non-invasive Treatments: First line treatment for
lymphedema, involves two phases: the reduction
therapy and the maintenance therapy. The first one
involves Complete Decongestive Therapy (CDT)
which is administered by a certified lymphedema
therapist, whose goal is to decrease symptoms and
limb volume. It is individualized for each patient, but
it typically includes Manual Lymphatic Drainage
(MLD), aerobic exercise, skin care, patient education
and compression strategies; such as using an
advanced decongestive pump and compression
garments.
Pneumatic devices used in CDT are often heavy
and can cause discomfort to the patient; who has to use
the devices at least twice per week for 50 minutes per
session (Kayõran et al., 2017). Once minimized limb
volume is achieved with CDT, after several weeks,
maintenance therapy begins. This may include self or
caregiver-administered MLD, compression garments,
exercise, wound prevention, and skin care. (Gillespie,
2018, Shaitelman, 2015)
While non-invasive treatment remains the
standard of care for BCRL, surgical management is
another avenue to treat persistent lymphedema,
particularly for patients who do not respond to non-
invasive treatments. Lymph Venous anastomosis
(LVA) is one such method but they are out of scope
of this paper.
2.2 Clinical Motivation: The Patient
Perspective
Aside from the long-term physical problems,
lymphedema also causes serious psychological and
social problems for patients. Affected women may
have decreased self-confidence resulting from a
distorted body image and negative emotions are often
reported including anxiety, frustration, sadness, anger,
fear, and increased self-consciousness (Taghian,
2014). As a result, in the last decade there is an
increased focus on the quality of life (QoL) and Patient
Reported Outcome Measures (PROMs). Most of the
Arm Symptom-Specific Questions in Questionnaires
on Quality Of Life as well as the ICHOM Breast
Cancer Reference guide refer pain, heaviness,
swelling/ tightness, loss of strength,
tingle/burning/pins and skin problems, elevation of
the arm and movement difficulties and the ability of
wearing clothes of choice as major everyday problems
(Cornelissen, 2018 and ICHOM Breast Cancer Data
Collection Reference Guide, 2017)
The necessity to wear compression sleeves are
viewed as barriers to daily life and the current
products are described as “terrible daily armour", one
sized, uncomfortable, tight, affecting clothes wearing
and patients’ discomfort is described by adjectives
such as “aching,” “heaviness,” and “pulling” (Hayes
et al., 2009, O'Toole, 2013). Numbness, dehydration
with chapping or sweating with itching, and risk of
bacterial or fungal infection are the most frequently
mentioned problems concerning the skin because of
the absence of pH-neutral moisturizer and
antibacterial layer in existent devices (Buragadda,
2015).
We had the chance to work closely with a 24-year-
old cancer survivor, who is a representative of the
Young Cancer Patient Association Sweden (Ung
Cancer). The association was founded in 2010 and
supports young cancer patients and survivors, aged
between 16-30 years old, as well as young people who
have a relative suffering or has died from cancer.
Through several discussions and a presentation by
patient representative, we were able to better
understand some everyday challenges that the young
cancer patients are facing. Given the relationship that
we had developed and our interest in the patients’
perspective we kindly asked the representative to
conduct a small research through the association
concerning breast cancer survivors who are suffering
from lymphedema. The research included two
questions: ‘’Describe your feelings and challenges
living with lymphedema’’ and “Would you use the
device we have designed’’.
Responses received from the community to the
first question were all arguing with the findings from
the literature while answers concerning our solution,
were all positive. Participants highlighted the need for
something innovative.
ClinMed 2020 - Special Session on Designing Future Health Innovations as Needed
324

2.3 Lymphedema Market Overview
and Economic Burden
According to the first overview of reported direct and
indirect patient-borne as well as society-borne costs
specifically associated with the treatment of BCRL in
literature; BCRL imposes a substantial economic
burden which is often neglected. Direct costs include
medical and therapeutic consultations, compression
garments, medications etc., while indirect costs refer
to productivity losses, values of lost income, unpaid
help and lost unpaid work. Mean direct costs per year
borne by patients ranged between USD$2306 and
USD$2574. Indirect costs borne by patients ranged
between USD$3325 and USD$5545 per year. Mean
direct treatment costs after 1 year of CDT ranged
between
€799 (= USD$1126.60) and USD$3165 (De
Vrieze, 2019). This amount of money mainly depicts
what is happening outside the EU and it is difficult to
be transferred. However, it remains a strong evidence
of BCRL economic burden. Therefore, we need more
high-quality, standardized health economic analyses
in order to have a clear view of the EU landscape and
push to acquire reimbursement.
There is only 1 industrial company per 10 million
that is dedicated to lymphedema products in Europe
and developing countries are virtually depleted of
industrial companies in this field (Schulze, 2018). At
the same time Alibaba.com offers 3390 lymphatic
drainage air pressure machine products.
We have conducted a small market research to
identify some of our direct competitors. FlexiTouch,
4 Chamber Arm Garment and LymphaPress have
been identified as the major ones. FlexiTouch, Tactile
System Technology, Minneapolis (USA,North
America), has an FDA approval and numerous
garment and program options but needs a continuous
connection to a large external device, it has a non-
ergonomic size and non-thermoregulated garment
and it costs around 1000€. 4 Chamber Arm Garment,
BioCompression Systems, NewJersey (USA,North
America), has similar advantages with FlexiTouch.
However, the cost varies between 914-3.450€ while
the external device has an outdated design, the
garment is made of an uncomfortable material and
there is an unreliable cable connection. Finally,
MegoAFEK Ltd., Afek (Israel, Middle East) is the
world’s leading innovator in pneumatic compression
therapy. LymphaPress is considered the leader in
lymphedema and venous insufficiency treatment and
it has strong clinical evidence with peer-reviewed
publications. Although it has proven positive results
its use is limited because of the large in size garment,
its heavy weight, lots of wire connection cables and
lack of patients’ mobility.
After listening carefully patients’ needs and
taking in mind the current market solutions, we have
concluded that we need a solution that is more
compact and non-dependent to external devices in
order to reduce fatigue, increase independence and
self-confidence.
3 PROPOSAL OF AN
INNOVATIVE PRODUCT
3.1 Specifications for Solution
Our solution consists of a device called
‘LymphMotion’ which comprises of two factors that
can be used in conjunction:
A portable pneumatic compression device (PCD)
in the form of a sleeve, ‘LymphMotion’ which the
patient can use at home in place of going into a
specialized clinic for pneumatic compression
(Pressure therapy) or a therapist for manual lymphatic
drainage (MLD). The PCD sleeve provides a gentle
massage movement to facilitate lymph fluid away
from the affected limb.
A complimentary compression garment that is
infused with microencapsulated aloe vera and
antimicrobial nanoparticles, in order to reduce
dryness and prevent bacterial or fungal infection. The
compression garment is to be used daily while the
patient is awake and during exercise.
During the conception-development procedure of
LymphMotion we kept in mind the IEC 62366 MD-
Application of usability engineering to medical
devices.
Intended Use: Alleviation of lymphedema in breast
cancer survivors who have undergone mastectomy,
and prevention of the disease to progress to more
serious stages or develop more serious complications
such as infections or cancer.
Intended User: Breast cancer survivors, who have
undergone mastectomy (ages 30-80).
User Environment: At home during the day and
during exercise.
3.2 Description of LymphMotion
The portable pneumatic compression device (PCD),
LymphMotion consists of a sleeve that is worn over the
affected limb (such as the arm) that includes a network
of 3D printed pockets designated in zones that can
inflate and deflate via air pressure. The network of
pockets acts as individual massage actuators. One-
Design of an Innovative Medical Device to Improve Quality of Life in Lymphedema Patients
325
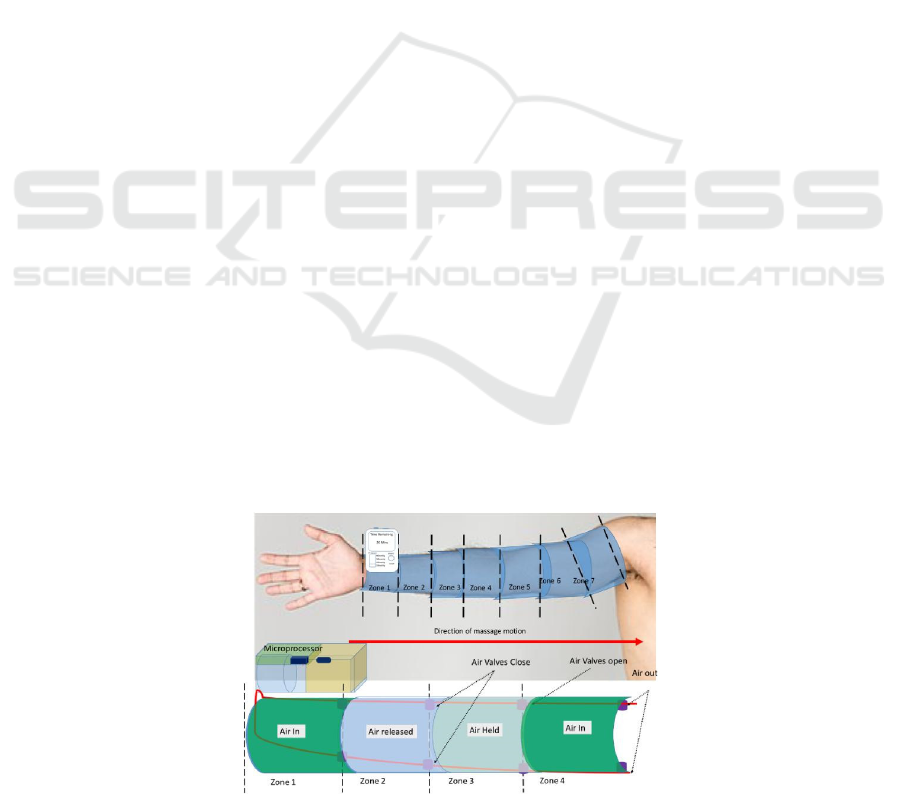
way air pumps and air valves which controls air flow
into the next zone of the sleeve in a controlled manner
using a microprocessor, in the direction of lymphatic
drainage. Air flow enters from the most distal point of
the limb away from the body (e.g. wrist on the arm)
and is released at the end of the sleeve closest to the
body – this direction ensures that the lymph fluid is
being drained away from the limb into the blood
circulatory system and to be eliminated via natural
processes (i.e. urination). A schematic of this can be
found in Figure 1. The device would be made in
different sizes to accommodate the patient.
Silicone can be used to make the pocket as it is a
low-cost, soft, durable material with a high elastic
modulus and power to weight ratio. They make safe
actuators for direct human interaction owed to their
lightweight and lack of rigid parts (Al-Fahaam et al.,
2018). This network of silicone pockets can be
manufactured using 3D printing methods. One such
method is a technique called Rapid Liquid Printing
(RLP) (Hajash et al., 2017; Papadopoulou et al.,
2017).
A piezoelectric pressure sensor that provides a
feedback loop to the microprocessor is used to ensure
the correct amount of pressure (mmHg) – depending
on which stage of lymphedema the patient is at, is
used during the massage on the patient, preventing
insufficient or dangerous levels of applied pressure
(Elwenspoek and Wiegerink, 2001; Meyer et al.,
2006). Gyroscope sensors are used to detect whether
the movement of the device is due to patient
movement or device inflation; this allows the user to
move while wearing the device while not
compromising on applied pressure; making the
device portable. The device can be powered by
rechargeable Lithium ion (Li-ion) batteries, with a
lifetime of 2000 cycles – enough cycles to last
approximately 5.5 years (Coyle et al., 2010;
Gorlatova et al., 2014).
3.3 Description of Compression
Garment
Compression garments are used by patients daily
from the first moment they wake up, for the duration
of the day, and during exercise. A common
complication of constantly wearing compression
garments is risk of bacterial and fungal infection that
results in dermatitis, or if there is a wound in the skin,
wound infections. Another issue is that compressive
garments can be uncomfortable and result in dry,
itchy, cracking skin (Lim and Davies, 2014; Vignes
and Arrault, 2009; Xiong and Tao, 2018). The
LymphMotion compression garment aims to reduce
these issues with current compressive garments in the
market.
The complimentary LymphMotion compression
garment (CG) addresses these two problems by
incorporating Anti-bacterial nanoparticles (ABNPs)
and encapsulated aloe vera into the fabric of the
compression garment. ABNPs exhibit antibacterial
and anti-inflammatory effects that can significantly
inhibit bacterial growth and reproduction. The
integration of NPs in the fabric can reduce the risk of
wound infection or accelerate wound healing should
the skin be injured. Combinations of nanosilver,
chitosan (CS), tungsten, and copper have been used in
production of textiles that is antibacterial. Fibre
fabrication methods such pressurized gyration
(Illangakoon et al., 2017; Wang et al., 2017) can be
used to create such fibres. Aloe Vera (AV) is
stabilized and encapsulated by immersing the fabric
into an AV micro-emulsion and inducing ultrasonic
waves to encapsulate and load the AV extract onto the
fabric (Ghayempour, Montazer and Mahmoudi Rad,
2016).
3.4 Regulatory Aspects
Lymph Motion is an active, non-invasive, non-
Figure 1: Schematic of the proposed pneumatic compression device LymphMotion and how it works.
ClinMed 2020 - Special Session on Designing Future Health Innovations as Needed
326

implantable Medical Device (MD) that complies with
the EC Regulation 2017/745/EU of May 5, 2017 on
Medical Devices. According to Article 52, Section 6
and Annex VIII, Rule 9, Section 6.1 of the MDR
2017/745/EU the above-mentioned MD is considered
as Class IIa MD.
As our product includes software that collects
patients’ pressure data, we need to comply with the
GDPR (General Data Protection Regulation) EU
2016/679. In collaboration with a DPO (Data
Protection Officer) we will take action to reduce data
volume collected, the recipients of these data and
especially for the clinical studies part, reduce the
duration of the process. We will also need to comply
with ISO/IEC 27032:2012 Guidelines for
cybersecurity.
4 CONCLUSIONS
Lymphedema continues to be clinically and
economically underestimated. The current available
treatment is insufficient, and data on the improvement
of patients’ quality of life is missing if not negative.
The burden is physical, social and psychological and it
is these dimensions that our solution tries to cover.
To bring our idea further, we will need funding to
create a prototype. In addition, according to the MDR
2017/745/EU and the new requirements on the Clinical
Evaluation of a Medical Device, in order to
demonstrate safety and performance requirements of
the device when used in accordance with the intended
purpose provided by the manufacturer, we will need to
proceed with biocompatibility and usability studies and
if the results are positive we should continue with a
Clinical Investigation. Further issues that would need
to be addressed would be the challenge of developing
Lymphmotion on an industrial scale. A business model
canvas is under development. Further options are under
consideration.
ACKNOWLEDGEMENTS
We would like to extend our thanks to Dr. Maria
Hagglund for hosting the immersive experience at
Uppsala University Hospital as well as the cancer
specialists for their invaluable knowledge. We thank
Emilya Bislimova, and the Young Cancer Patient
Association Sweden for their patient perspectives. We
also thank our teammates at the 2019 EIT Clinmed
Summer School Tiberiu Niță Mihai and Marta Tallon.
REFERENCES
Al-Fahaam, H., Davis, S., Nefti-Meziani, S., Theodoridis, T.,
2018. Novel soft bending actuator-based power
augmentation hand exoskeleton controlled by human
intention. Intell. Serv. Robot. 11, 247–268.
https://doi.org/10.1007/s11370-018-0250-4
Brahma, B., Yamamoto, T., 2019. Breast cancer treatment-
related lymphedema (BCRL): An overview of the
literature and updates in microsurgery reconstructions.
Eur. J. Surg. Oncol. 45, 1138–1145. https://doi.
org/https://doi.org/10.1016/j.ejso.20 19.01.004
Buragadda S, Alhusaini AA, Melam GR, Arora N.,2015.
Effect of complete decongestive therapy and a home
program for patients with post mastectomy lymphedema.
J Phys Ther Sci. 2015;27.
Cemal, Y., Pusic, A., Mehrara, B.J., 2011. Preventative
Measures for Lymphedema: Separating Fact from
Fiction. J. Am. Coll. Surg.213,543–551.
https://doi.org/https://doi.org/10.1016/j.jamc ollsurg.20
11.07.001
Cornelissen AJM, Kool M, Keuter XHA, Heuts EM,
Piatkowski de Grzymala AA, van der Hulst RRWJ, Qiu
SS. 2018.Quality of Life Questionnaires in Breast
Cancer-Related Lymphedema Patients: Review of the
Literature. Lymphat Res Biol. 2018 Apr;16(2):134-139.
Coyle, S., Lau, K.T., Moyna, N., O’Gorman, D., Diamond,
D., Di Francesco, F., Costanzo, D., Salvo, P., Trivella,
M.G., De Rossi, D.E., Taccini, N., Paradiso, R., Porchet,
J.A., Ridolfi, A., Luprano, J., Chuzel, C., Lanier, T.,
Revol-Cavalier, F., Schoumacker, S., Mourier, V.,
Chartier, I., Convert, R., De-
Moncuit, H., Bini, C., 2010. BIOTEXBiosensing textiles for
personalised healthcare management. IEEE Trans. Inf.
Technol. Biomed.14,364–370. https://doi.org/10.1109/TI
TB.2009.2038484
De Vrieze, T., Nevelsteen, I., Thomis, S., De Groef, A.,
Wiebren A. A. Tjalma, Gebruers, N., Devoogdt, N.,
2019. What are the economic burden and costs
associated with the treatment of breast cancer-related
lymphoedema? A systematic review. Supportive Care in
Cancer https://doi.org/10.1007/s00520-019-05101-8
DiSipio T, Rye S, Newman B, et al. 2013. Incidence of
unilateral arm lymphoedema after breast cancer: A
systematic review and meta-analysis. Lancet Oncol
2013;14:500-15.
Douglass, J., Kelly-Hope, L., 2019. Comparison of Staging
Systems to Assess Lymphedema Caused by Cancer
Therapies, Lymphatic Filariasis, and Podoconiosis.
Lymphat. Res. Biol.17,550–556. https://doi.org/10.1089/
lrb.2018.0063
Elwenspoek, M.C., Wiegerink, R.J., 2001. Mechanical
microsensors, Microtechnology and MEMS, ISSN 1615-
8326. Springer.
Ghayempour, S., Montazer, M., Mahmoudi Rad, M., 2016.
Simultaneous encapsulation and stabilization of Aloe
vera extract on cotton fabric for wound dressing
application. RSC Adv. 6, 111895–111902.
https://doi.org/10.1039/C6RA22485G
Design of an Innovative Medical Device to Improve Quality of Life in Lymphedema Patients
327

Gillespie, TC., Sayegh, H.E., Brunelle, C.L., Daniell, K.M.,
Taghian, AG., 2018. Breast cancer-related lymphedema:
risk factors, precautionary measures, and treatments.
Gland Surg. 2018 Aug;7(4):379-403.
Gorlatova, M., Sarik, J., Grebla, G., Cong, M., Kymissis, I.,
Zussman, G., 2014. Movers and Shakers: Kinetic Energy
Harvesting for the Internet of Things, in: The 2014 ACM
International Conference on Measurement and Modeling
of Computer Systems, SIGMETRICS ’14. ACM, New
York, NY, USA,pp.407–419. https://doi.org/10.1145/
2591971.2591986
Hajash, K., Sparrman, B., Guberan, C., Laucks, J., Tibbits,
S., 2017. Large-Scale Rapid Liquid Printing. 3D Print.
Addit. Manuf. 4, 123–132. https://doi.org/10.1089/
3dp.2017.0037
Hayes, S.C., Reul-Hircher, H., Turner, J., 2009. Exercise and
Secondary Lymphedema: Safety, Potential Benefits, and
Research Issues. Med. Sci. Sport. Exerc. 41.
Ichom Breast Cancer Data Collection Reference Guide
Version 1.0.1 Revised: March 30, 2017
IEC 62366-1:2015Medical devices Ñ Part 1: Application of
usability engineering to medical devices, 2015, 1
st
Edition
Illangakoon, U.E., Mahalingam, S., Wang, K., Cheong, Y.-
K., Canales, E., Ren, G.G., Cloutman-Green, E.,
Edirisinghe, M., Ciric, L., 2017. Gyrospun antimicrobial
nanoparticle loaded fibrous polymeric filters. Mater. Sci.
Eng. C 74, 315–324. https://doi.org/https://doi.org/
10.1016/j.msec. 2016.12.001
Kayõran, O., De La Cruz, C., Tane, K., Soran, A., 2017.
Lymphedema: From diagnosis to treatment. Turkish J.
Surg. 33, 51–57. https://doi.org/10.5152/turkjsurg.
2017.3870
Keast, D.H., Despatis, M., Allen, J.O., Brassard, A., 2015.
Chronic oedema/lymphoedema: Under-recognised and
under-treated. Int. WoundJ.12,328–333. https://doi.org/
10.1111/iwj.12224
Lim, C.S., Davies, A.H., 2014. Graduated compression
stockings. C. Can. Med. Assoc. J. = J. l’Association
medicale Can. 186, E391-8. https://doi.org/
10.1503/cmaj.131281
ltd, R. (2019). Global Compression Garments Market Size,
Market Share, Application Analysis, Regional Outlook,
Growth Trends, Key Players, Competitive Strategies and
Forecasts, 2018 To 2026. [online]
Researchandmarkets.com. Available at: https://www.
researchandmarkets.com/reports /4760532/global-
compression-garments-market-size-market [Accessed
26 Nov. 2019].
Meyer, J., Lukowicz, P., Troster, G., 2006. Textile Pressure
Sensor for Muscle Activity and Motion Detection, in:
Proceedings - International Symposium on Wearable
Computers, ISWC.pp.69–72. https://doi.org/10.1109/
ISWC.2006.286346
O'Toole J, Jammallo LS, Skolny MN, et al.2013.
Lymphedema following treatment for breast cancer: a
new approach to an old problem. Crit Rev Oncol
Hematol.
Papadopoulou, A., Laucks, J., Tibbits, S., 2017. From Self-
Assembly to Evolutionary Structures. Archit. Des. 87,
28–37.
Regulation (EU) 2017/745 of the European Parliament and of
the Council of 5 April 2017 on medical devices,
amending Directive 2001/83/EC, Regulation (EC) No
178/2002 and Regulation (EC) No 1223/2009 and
repealing Council Directives 90/385/EEC and
93/42/EEC, OJ L 117, 5.5.2017, p. 1–175
Regulation (EU) 2016/679 of the European Parliament and of
the Council of 27 April 2016 on the protection of natural
persons with regard to the processing of personal data and
on the free movement of such data, and repealing
Directive 95/46/EC (General Data Protection
Regulation), OJ L 119, 4.5.2016, p. 1–88
Rockson, S.G., 2018. Lymphedema after Breast Cancer
Treatment. N Engl J Med. 2018 Nov 15;379(20):1937-
1944
Schulze, H., Nacke, M., Gutenbrunner, C., Hadamitzky,
C.,2018. Worldwide assessment of healthcare personnel
dealing with lymphoedema. Health Econ Rev.
2018;8(1):10.
Shaitelman, S.F., Cromwell, K.D., Rasmussen, J.C., et al.
2015. Recent progress in the treatment and prevention of
cancer-related lymphedema , CA Cancer J Clin.
Shih, Y.-C.T., Xu, Y., Cormier, J.N., Giordano, S., Ridner,
S.H., Buchholz, T.A., Perkins, G.H., Elting, L.S., 2009.
Incidence, Treatment Costs, and Complications of
Lymphedema After Breast Cancer Among Women of
Working Age: A 2-Year Follow-Up Study. J.
Clin.Oncol.27,2007–2014. https://doi.org/10.1200/JCO.
2008.18.3517
Taghian, N.R., Miller, C.L., Jammallo, L.S., O’Toole, J.,
Skolny, M.N., 2014. Lymphedema following breast
cancer treatment and impact on quality of life: A review.
Crit. Rev. Oncol. Hematol. 92, 227– 234.https://doi.org/
https://doi.org/10.1016/j.c
ritrevonc.2014.06.004
Vignes, S., Arrault, M., 2009. Effets Indésirables de la
Compression/Contention dans le Traitement des
lymphÏdèmes des membres. J. Mal. Vasc.34,338–345.
https://doi.org/https://doi.org/10.1016/j.jmv. 2009.07.00
1
Wang, L., Hu, C., Shao, L., 2017. The antimicrobial activity
of nanoparticles: Present situation and prospects for the
future. Int. J. Nanomedicine Volume 12, 1227–1249.
https://doi.org/10.2147/IJN.S121956
Xiong, Y., Tao, X., 2018. Compression Garments for
Medical Therapy and Sports. Polymers (Basel). 10.
https://doi.org/10.3390/polym10060663
ClinMed 2020 - Special Session on Designing Future Health Innovations as Needed
328
