
Adding Value to Translational Informatics through the Semantic
Management of Drug to Drug Interaction
Radmila Juric
a
University of South Eastern Norway, Kongsberg, Norway
Keywords: Translational Informatics, OWL/SWRL, Drug-to-Drug Interactions.
Abstract: Translational informatics, aimed at bridging the gap between biomedical scientific knowledge and clinical
practice has changed the way we use rapidly growing information from biomedical research and bring it closer
to clinical practice. Software technologies play an important role in this process, particularly if they help in
understanding and manipulating the meaning of data and information generated in biomedical research and
translate it into semantic suitable for clinical practice. In this paper, we propose software architectural and
conceptual computational models, which use semantic technologies in order to explore the meaning of the
relationships between drugs when they interact in clinical practice. The data about drug to drug interactions,
available from biomedical research, is reusable in instances where they are decisive factors in drug
administration in clinical practice. We explore the power of semantic web technologies and SWRL enabled
OWL ontologies to demonstrate the applicability and feasibility of our proposal in translational informatics.
1 INTRODUCTION
This research explores the personalization of patient
medication lists in terms of finding potential drug
interactions, because of the combination of prescribed
drugs. We are interested in drug to drug interactions
(DDI) which may appear if the therapeutic effect of
one drug changes because of the presence of another.
This problem is not easy to resolve for many reasons.
One of them is that the solution might require a
synergy of knowledge and expertise across the
disciplines of biomedical science, clinical practice
and computer science. Modern medicine strives for
personalization, hoping to include gender differences
and the clinical physiological effect drugs may have
on an individual patient, because every patient is
different. However, we still do not perform clinical
trials which take this personalization into account.
Furthermore, the advances of knowledge discoveries
in biomedical science, are not fast-forwarded to
clinical practice and the gap between the two is
widening. There are examples where translational
bioinformatics (Tsafnat et al., 2013) may address the
problem, but a long-term solution which addresses a)
data sharing between biomedical science and clinical
a
https://orcid.org/ 0000-0002-0441-0694
practice and b) personalization of patient medication
lists to avoid potential DDI, might be the only way
forward. However, a) and b) above are interwoven. If
we share the data between biomedical research and
clinical practice, we will find more about DDI. In
order to align personalized medication lists, for the
purpose of eliminating potential DDI between the
drugs in the list, we would need more than just data
sharing. We would need to understand the semantic
relationship between clinical recommendations, i.e.
prescribed drugs, drug therapeutic targets
(protein/genes) and related biological functions, in
the context relevant to a patient. If there is another
drug, which shares the same or similar therapeutic
target, intentionally or un-intentionally, then these
two drugs could have a variety of interactions, which
should be semantically explained. DDI would depend
on the exact involvement of each drug in their
therapeutic targets/biological functions of the patient.
Software engineering solutions, with data sharing
across disciplines, and reasoning upon the collected
data, in order to find potential DDI for the patient’s
medication list, would require a software architectural
(SA) model first, which specifies sources of shared
data and computational models for identifying
relevant DDI. A software application, created from
268
Juric, R.
Adding Value to Translational Informatics through the Semantic Management of Drug to Drug Interaction.
DOI: 10.5220/0009376002680275
In Proceedings of the 13th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2020) - Volume 3: BIOINFORMATICS, pages 268-275
ISBN: 978-989-758-398-8; ISSN: 2184-4305
Copyright
c
2022 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved

the SA would work for patient/clinicians and secure
the best possible medication lists, personalized in a
particular context. The same application should allow
updating the semantics of the drug
1
-target-drug
2
relationship from biomedical research and thus fast
forward biomedical knowledge, on discovered DDI,
towards creating a personalized medication list.
Our proposal uses Semantic Web Technology
(SWT) and its languages OWL/SWRL
(OWL/SWRL) for defining the reasoning process
upon data shared from biomedical experiments, with
drug
1
-target-drug
2
pathways and patent clinical data,
i.e. medication lists. We infer DDI relationships
between the two drugs, from the patient medication
list, through semantic reasoning upon the data which
originate in biomedical research and clinical practice.
The paper is organized as follows. Related Work
lists examples of finding DDI, using software
solutions with Natural Language Processing (NLP),
semantic technologies and reasoning. Our proposal,
described in the sections, which follow, gives a
reusable SA model which hosts computations based
on reasoning upon shared biomedical and clinical
data. We illustrate the reasoning process and debate
the proposal in the Implementation and Conclusions.
2 RELATED WORK
Biomedical research has advanced significantly and it
is almost impossible to systemize results of research
advances and create an overall picture of new
knowledge which is emerging as we speak. In the
DDI field, we can go back through decades and find
a variety of research publications which highlighted
the problem. In this section we chose a selection of
interesting papers which either influenced us, or
illustrate new ideas to finding DDI.
The authors of (Herrero-Zazo et al., 2013) use
NLP, which is still very popular for retrieving textual
information from biomedical sources and finding
DDI. In order to improve NLP they created an
annotated corpus of pharmacological substances and
DDI, sourced at DrugBank database (Knox, 20|11)
and 233 Medline abstracts. The authors from (Aywaz
et al. 2015) created a complete data set of DDI
information from 14 public sources and merged them,
but found that there are inconsistencies and
overlapping between sources which disseminate
information on DDI. In (Segura-Bedmar et al.m
2010) linguistic extraction techniques and a hybrid
linguistic approach to DDI detection are used, that
combine shallow parsing, provided by UMLS
(UMLS, 2009) tools, and syntactic simplification
with pattern matching. Lexical patterns achieved
reasonable precision. In (Liu et al., 2019) the
detection of adverse drug events (ADE) from social
media is shown. This is not exactly DDI, but it is a
refreshing way of getting information fast and
exploring social media for learning differently about
DDI. Because of the type of sources used in this work,
a semi-supervised learning method, with weighted
features is used to distinguish between ADE and non-
ADE. (Kim et al., 2015) use text mining techniques
to identify DDI in the body of unstructured medical
text and consequently, a support vector machine with
a linear kernel is a good option for the task. The
authors of (Xhoua, et al., 2018) use a position aware
and multi-task deep learning to extract DDI from
unstructured medical texts. Deep neural networks,
which use words and their positions in the
unstructured text, for defining latent features and thus
avoiding explicit feature engineering, for finding
DDIs are in (Sahu and Anand, 2018).
All these examples show that NLP, information
extraction, text mining and statistical classifications,
with learning technologies, dominate the research
scene for one important reason. The Semantic of the
DDI from biomedical research is often buried in
unstructured biomedical texts.
Ontologies are not often used for detecting DDIs.
If they appear in research, they are mostly controlled
vocabularies for cumulating knowledge as results of
reasoning. Data retrievals are carried out with
SPARQL. Drug Interaction Ontology from
(Yoshikawa, 2004) is a very old, but formal ontology,
which accumulates reusable knowledge in molecular
pharmacology. Its information model is based on
fundamental concepts of biological interactions. The
paper was published before the standardization of the
SWT and its languages and therefore it can not be
compared with modern SWT solutions. Potential
DDI are results of retrievals upon ontological
concepts. The authors of (Alhaj et al., 2019) created
the ontology identifying DDI, but reasoning classifies
only DDI effects: reduction, synergism and toxicity.
In (Saleha et al, 2017) the DDI ontology helps in drug
discovery investigations, in (Sara et al., 2018) a drug
interaction ontology contains information about
ADE, and in (Grando et al., 2012) ontology is used
for safe and effective generic prescription principles.
DINTO ontology, which contains formal
representation of different types of DDI is available
in (Herrero-Zazo, 2015). It is very complex and
models DDIs as classes and properties. This may have
an impact on the OWL model’s efficiency and
reusability. It is interesting that they created a set of
inference rules for a variety of DDIs. They are
Adding Value to Translational Informatics through the Semantic Management of Drug to Drug Interaction
269

inferred on the basis of their pharmacological
mechanisms, which in turn depend on the biological
process leading to their occurrence. These interwoven
facts must be presented within the ontology, possibly
through chaining of object properties. This can lead
to inefficiency in the reasoning process, if the object
properties chaining were to be a part of any stand-
alone software application. If DINTO were used as a
formal vocabulary, then its efficiency in real life will
depend on the way SPARQL performs, and not on the
constraints, imposed by OWL object properties.
We could not find any solutions, which would
infer DDI from prescribed medication lists.
However, DINTO ontology could be used in our
proposal as a supporting source of information for
understanding the context within which we infer DDI
for a particular patient and his/her medication list.
There is only one publication which comes closer
to this research than any other. It uses GalenOWL
ontology for drug recommendation discovery
(Doluaverakis et al., 2003). It is an online-service,
based on queries aimed at drug-to-drug and drug-to-
disease discoveries. It offers conceptual reasoning
rules upon a set of domain ontologies for inferring
OWL properties and thus recommends a particular
drug to a patient. Its rule base engine evaluates
conflicts between drug therapeutic indications and
contraindications and thus indicates implicitly, but
not explicitly the problem of DDI.
3 THE PROPOSAL
The proposal consist of two parts. In the first part we
introduce the SA model which specifies the main
software components of the layered and component
based architectural style. The model is a prescription
on how to build software applications for delivering
the inference of potential DDIs.
In the second part, we look at the specificity of the
computational model from the software application
perspective and define OWL classes, with their
individuals and properties, and the reasoning process.
However, before we define the proposal, we
briefly debate the role of SWT in this problem
domain. This is important because we do NOT create
another formal ontology in biomedical science.
SWRL enabled OWL ontologies are here to (i)
become a part of a software engineering application
(ii) exploit the semantic relevant for the task of
discovering DDI and (iii) contribute towards a
computational model, which detects a potential DDI
and change the proposed medication list, if necessary.
3.1 Why SWT?
SWT and its layered cake has widely been used, since
its standardisation in 2004, for interpreting the
meaning of data available on the Web. It is perfect
for building common ontologies and controlled
vocabularies across domains, enriched with reasoning
rules in SWRL, and thus bringing inference and more
semantics to the Web. We can represent knowledge
with OWL/SWRL because of its powerful
representation through description logic.
There are numerous possibilities of using and
exploiting SWRL enabled OWL ontologies, but we
would like to emphasise its use outside the Web and
controlled vocabularies, i.e. knowledge-bases for
many reasons. The most important reason is that
computational models, which house SWRL enabled
reasoning upon OWL concepts, bring inference
without either having complex knowledge systems in
the background or using Artificial Intelligence (AI)
algorithms for creating inference. Therefore we talk
about software engineering applications of the SWT
technology. We can also create SWRL/OWL
inference on an ad-hoc basis and address constant
changes in environments we model. If we add that
SWRL reasoning upon OWL concepts can be
teamed-up with filtering, ranking, tagging, semantic
annotations, transactional and big data processing,
and performing prediction analytics (Juric 2016),
(Juric and Kim 2017) then our proposed software
engineering solution, outside formal ontologies and
knowledge-bases, is a promising start.
3.2 Software Architectural Model
The SA model is proposed in Figure 1. Its middle
vertical content houses a computational model which
performs reasoning upon SWRL enabled OWL
ontologies. This repository contains semantics of
both: drug(s) specifications, their therapeutic targets
(genes/proteins), and biological functions and
clinical-level of physiological effects, relevant to the
prescribed medication list (for a particular patient).
Therefore prescribed medication list originates in the
software application which Manipulates Clinical
Data (right vertical part of Fig 1.)
The left part of Fig. 1 illustrates repositories from
the biomedical (BM) field and may include BM
Databases {BMDB
1
, BMDB
2
,, …BMDBn}, existing
BM ontologies {BMOnt
1
, BMOnt
2
,, …BMOnt
m
}
containing results of BM research. Drug
specifications, explanations on their therapeutic
targets, and knowledge of existing DDI are all
BIOINFORMATICS 2020 - 11th International Conference on Bioinformatics Models, Methods and Algorithms
270

available within BM software applications, which
Manipulates Biomedical Data, and their repositories.
The right side of Figure 1 (Manipulate Clinical
Data) contains data/software applications specific to
clinical data and patients Electronic Health Records
(EHR). Patient medication lists may be generated
there (not compulsory) and therefore the clinical level
of physiological effects of prescribed drugs to the
patient, might also be known within this environment.
The proposed computational model sits between
the two environments (left and right part of Figure 1)
and Creates Personal DDI. It bridges the gap
between the knowledge available from BM research
and data related to clinical practices. The data sharing
between these two environments, which are in reality
two separated worlds, is essential if we wish to bridge
the gap. DDI OWL Ontology with SWRL, can be
populated with the semantic of data from both sides
(Juric, 2016). This is the only way we can perform
inference where the reasoning creates a DDI on an ad-
hoc basis.
3.3 Semantic of DDI Relationship
The section on Related Work shows various ways of
describing the semantics of potential DDI. One
publication proved to be extremely beneficial for
performing reasoning upon biomedical concepts in
general. The relationship between drugs and their
therapeutic targets, described through semantic
predications, related to all medications, available in
Sem-MedDB (Kilicogly et al., 2012) is almost ideal
for being converted into OWL ontologies.
Semantic PREDICATES are defined in the
UMLS Manual (UMLS) (Ahlers et al., 2007), and the
authors of (Zhang et al., 2014) also use them as an
input into their own method of finding DDIs. These
predications are very attractive to software engineers
because they are TRIPLETS:
subject-PREDICATE-object
where subject is a particular drug and object is its
target (genes/enzymes). DDI between two drugs can
be identified, if we have the following logic:
subject
i
-PREDICATE
j
-object
k
-PREDCATE
l
-
subject
m
where subject
i
is NOT the same as subject
m
(these are
two different drugs) and therefore
subject
i
≠ subject
m
→ DDI
i,j
Figure 1: Software Architectural Model.
This means that if we have two drugs, both involved
in the same target (genes / enzymes / biological
function) through different predicates, and thus would
interfere with each other. The types and number of
PREDICATES, which can be extracted from the
SemMedDB and UMLS Manual are ideal for logic
reasoning. We can transfer scientifically agreed
predications directly into OWL (object properties).
This is one of the most important task when creating
a reasoning process using OWL/SWRL: semantically
rich object properties. There are many predicates in
SemMedDB, such as TREATS, AFFECTS,
INTERACTS_WITH, STIMULATES, INHIBITS,
which can be object properties in OWL. They can
connect individuals between drug and target concepts
in drug-tehrapeutic target pathways.
Figure 2: DDI
i,j
is defined between any pair of drugs.
Figure 2 shows a possible DDI between two drugs
Drug
i
and Drug
j
from the set of n drugs D= {Drug
1
,
Drug
2
,, ..Drug
n
}, where we allow up to n-1 potential
DDI between them. Some of DDI
j,k
might be
imported from repositories of the BM research (left
part of Fig. 1) and some might be known to clinicians
(right part of Fig 1). If there is no knowledge about
{BMOnti,… BMOntom}
OWL
DDI OWL Ontology Clinical
with SWRL Practice
Drug
i
Drug
j
Gene/
Target
D
D
I
i
,j
Pi
Pj
Manipulate
Biomedical
Semantics
Create
Personal
DDI
Manipulate
Clinical
Data
OWL - API
HER
Med.
Lists
Report
{BMDB1,BMDB2,. BMDBn}
Adding Value to Translational Informatics through the Semantic Management of Drug to Drug Interaction
271

potential DDIi,j we have an opportunity to infer it
through reasoning if the semantics of subject
i
-
PREDICATE
j
-object
k
-PREDCATE
l
-subject
m
allows. Broken one directional lines in Fig. 2 indicate
therapeutic pathways for a drug, and the solid line
denotes (potential DDI). Abbreviation Pj and P
l
is for
two particular PREDICATES.
3.4 Semantic Relationship between
Drugs and Their Targets
We identify examples of predicates which can be
used for illustrating the semantic between Drugs and
their targets in order to define an OWL model.
Let us assume that the relationship between Drug
i
,
and gene is defined as a triplet
(Drug
i
-PREDICATE
j
-Gene)
where Drug
i
affects a particular Gene. However, the
same Gene might be involved, as a target, with
Drug
m
. This would require the definition of another
PREDICATE, between the same Gene and Drug
m
. If
we put these two triplets together,
Drug
i
-PREDICATE
j
-Gene-PREDCATE
l
-Drug
m
then the above construct can create Fig. 3, which
reads: Drug
i
AFFECTS a particular gene, but this
gene might be INVOLVED IN another Drug
m
.
Fig. 3 sends two messages. If there is a predicate
AFFECTS for an approved target for Drug
i
(black
arrow) we investigate if there is a potential predicate
INVOLVED-IN for the same target, but for a known
or approved Drug
m
(red arrow) The semantic of
predications is read in both directions. We may know
that a particular gene is involved in Drug
m
which
might require investigating is there is a drug Drug
i
which affects the same gene. From the computer
science perspective, we may allow multiple instances
of Drugi and Drugj, to appear in Fig. 3.
Figure 3: AFFECTS/INVOLVED-IN Predicates may be
used both ways.
3.5 Owl Model
One of the most important use of semantic
predication is in grasping semantic important for the
discovery of DDIs. From Figure 3, all
PREDICATIONS can be easily converted into OWL
constraints in ontological modelling. This means that
when building an ontological model we will be in a
position to use some PREDICATIONS as either
asserted OWL object properties or inferred.
In order to infer potential DDI between the two
drugs Drugi and Drugm, the most convenient way
would be to conceptualise them into two separate
OWL classes, which may change their role from
domain to range, to allow both way of reading form
Figure 3 (red and black arrows).
Figure 4: OWL Model derived from Figure 3.
In Fig 4. red and black arrows denote asserted
object properties (AFFECTS and INVOLVED-IN)
and the blue arrow is a new object property which is
inferred: It denotes a potential DDI
i,j..
Fig. 4 also shows a basic principle of conceptual
modelling of the OWL ontology: potential DDI
i,j
can
be inferred between Drug
i
and Drug
m
through
reasoning, if there existed a triplet Drug
i
-AFFECTS-
Gene-INVOLVED_IN-Drug
m
where Drug
i
is not the
same as Drug
m
. Fig. 5 shows a conceptual SWRL
rule with asserted PREDICATES are asserted from
SemMedDB for performing the reasoning.
Figure 5: Conceptual SWRL rule for Fig. 3 and 4.
The rule in Fig. 5 would work as long as semantic
predication is correct. Object properties affects (?x,
Gene and involved_in (?y,Drug
m
) may be replaced with any
other type of predicates available in SemMedDB or UMLS
and inferred using SWRL.
Drug
i
(?x) ˄ affects (?x, Gene) ˄ Gene(?y) ˄
involved_in (?y,Drug
m
) ˄ differentFrom (?x,?y)
Drug_Interaction (?x, ?,y)
Drug
i
Gene
Drug
m
AFFECTS
INVOLVED-IN
INVOLVED-IN
AFFECTS
Drug
i
Drug
m
Gen
e
BIOINFORMATICS 2020 - 11th International Conference on Bioinformatics Models, Methods and Algorithms
272
4 IMPLEMENTATION
The implementation of the middle part of the SA
model in Fig. 1 for Creating Personal DDI is based
on the reasoning process which conforms to Figures
3,4 and 5. The prototype was implemented as a Java
application, adopted from the research on biomedical
discoveries (Almami et al., 2016), (Almami et al.,
2017) and the modelling of semantic software
applications (Patadia et al., 2011), (Shojanoori, 203).
Due to space restrictions we show two important
aspects of the implementation.
Firstly, Fig. 6 illustrates a set of PREDICATES
extracted from public databases in order to test our
reasoning and the implementation.
Figure 6: Selection of Semantic Predications for the
Implementation.
We ran experiments for populating OWL classes
with individuals, extracted from the peer reviewed
papers on DDI discoveries. Figure 6 has data
extracted from Table 1 of (Zhang et al., 2014) and
thus we were able to define numerous triplets. We
also added a few other predicates available in some of
the sources described in the related work, which
shows that we can use any other set of biomedical
data (from the left part of the SA in Figure 1.) and
create more suitable triplets for our OWL model. In
cases where biomedical research results do not
generate knowledge in the format of predications, it
would not be difficult to create them either through
the software application from Figure 1 or by
exploring the nature of OWL object properties
defined Fig.4.
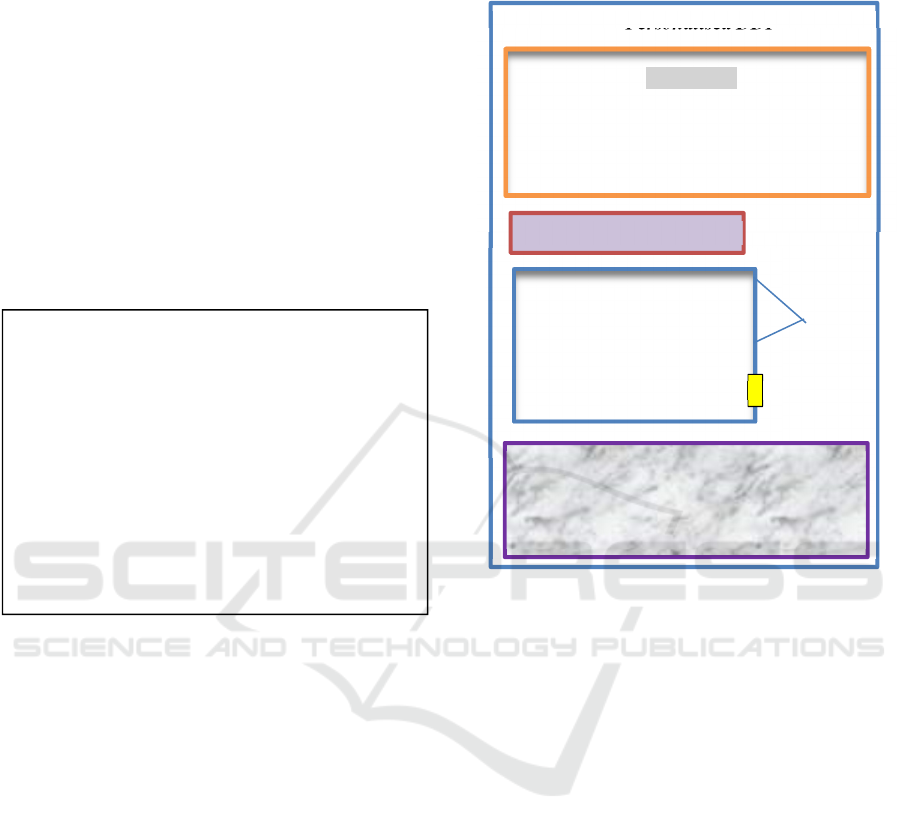
Secondly, an example of user interface for the
prototype is in Figure 1. From the patient medication
list, which initially contained Aspirin, Ibuprofen,
Warfarin, Diazepam, Lysinopril, Thyroxine, two
drugs have potentially known DDI: Ibuprofen and
Warfarin. However, our reasoning with SWRL
detected a new inferred DDI between Lisinopril,
Thyroxine.
Figure 7: Excerpt from Prototype UI.
However, our prototype, as an illustration of the
concept defined in Figure 1, which pushes forward
translational informatics, using efficient and light
weight software technologies, based on semantic
reasoning, shows only an excerpt of the overall
research. The comparison of two drugs, based on
semantic predications, uses bindings of drug
therapeutic compounds to intentional target genes.
There is much more semantic in biomedical science
which can enrich our OWL model and predications.
They include semantic of biological functions from
molecular to organism level to pathological function
related to diseases, to mention just a few.
5 CONCLUSIONS
Translational bioinformatics has come of age (Butte,
2008), (Machado et al., 2015), (Payne and Embi,
2015), and computational algorithms can be used for
assisting in experiments and analyzing results of
biomedical research at bio-molecular level.
However, we have not resolved all the problems we
identified more than a decade ago. The dissemination
Aspirin –INHIBITS -EGF-INTERACTS_WITH-
Ascorbic Acid
Lisinopril-INHIBITS-VIP-STIMULATES -
Thyroxine
Metformin -STIMULATES-Glu-
INTERACTS_WITH-Aspirin
Metformin-STIMULATES-THI-
INTERACTS_WITH -Pioglitazoone
Trastuzumab-INHIBITS-ld HER2 INHIBITS-
Pertuzumab
Ibuprofen-INHIBITS- COX-INTERACTS_WITH-
Warfarin
Nacfillin-INDUCES-CYP3A4-AFFECTS-Warfarin
Ibu
p
ro
f
en-INHIBIT
S
-
P
TGS52-INTERACT
S
-
Personalised DD
I
Patient John Smith
Diag. Arthritis,
Knee replacement
Hypothyroidism
GP James Brown
Ibuprofen
Paracetamol
Warfarin
Diazepam
Lisinopril
Thyroxine
DDI Warnings in Yellow
Known DDI are blue lines
Check the dosage
Medication List
Adding Value to Translational Informatics through the Semantic Management of Drug to Drug Interaction
273

of and sharing viomedicla research/data in lcinicla
applications is not common therefore translational
bioinformatics is still evolving. This research shows
that we can enhance it with the manipulation of
semantics of and reasoning from the results of
biomedical research, which in turn derives new
knowledge and tools in/for clinical practice and
medicine in general. This is an opportunity for all of
us to allow the synergy between biomedical and
clinical data and secure that clinical practices
encompass results from biomedical science, because
the gap between the two has not been closed.
This paper just touches the top of the iceberg of
opportunities we may have in the field of adding
value to translational informatics. There are
opportunities of reusing the conceptual model from
this paper for the whole range of problem domains,
from predicting side effects from a drug to therapeutic
targets relationships, to looking at un-intentional
binding of drugs and therapeutic targets, which could
help to define drug repositioning, to mention just a
few (Juric, 2019) (Juric and Almami, 2019).
Therefore this work continues towards the
development of software application, from the same
proposed SA from Figure 1, with reasoning upon
SWRL enabled OWL ontologies as a part of new
computational models, but in different parts of
biomedical fields. The only prerequisite is that data
sharing and dissemination of biomedical research is
essential for progressing in medical science.
For readers interested in methods of populating
OWL ontologies from databases and structured
repositories, which exist in biomedical research, we
suggest reading a few publications (Juric, 2019),
(Saaidi et al., 2010). For readers interested in
understanding how the SA from Fig. 1 can be
implemented as a software application, which
involves accessing OWL ontologies and computing
with SWRL through OWL–API, we suggest reading
(Juric, 2016), (Juric and Kim, 2016), (Tarabi and
Juric, 2018), (Shojanoori,, 2013).
For the full deployment of the SA from Figure 1,
in terms of commercialising the prototype, we face
expected obstacles such as
(i) gaining access to a variety of data banks,
databases and knowledge repositories from
biomedicine and
(ii) the acceptance of this type of applications in
clinical practice.
Resolving (i) and (ii) would require changes in the
way clinical trials are conducted and biomedical
research financed. However, it would also require
changes in the way we manage differences between
interests of pharmaceuticals and biomedical
scientists.
REFERENCES
Moore, R., Lopes, J., 1999. Paper templates. In
TEMPLATE’06, 1st International Conference on
Template Production. SCITEPRESS.
Smith, J., 1998. The book, The publishing company.
London, 2
nd
edition.
Ahlers, C.B., Fiszman, M., Demner-Fushman, D., Lang, F.
M.T. Rindflesch, C. 2007 Extracting Semantic
Predications from Medline, Citations for
Pharmacogenomics, Pacific Symposium on
Biocomputing 12:209-220(2007).
Alhaj, F., Obeid, N., Qutishat, D., Harahsheh, H. Al.,
Hammo B. 2019. Detecting DDI Using Ontology: Drug
Mechanism of Action in 2019 IEEE Jordan
International Joint Conference on Electrical
Engineering and Information Technology (JEEIT)
Almami, E. Almami, I., Juric, R. 2016. Knowledge
Dissemination In Biomedical Science: Using
Ontological Reasoning For The Analyses Of Activated
Enzymes In Various Diseases, In Proceedings of the
21st SDPS 2016 conference, December 2016
Almami, E., Juric, R. Almami, I. 2018. Biomedical
Semantics: Exploring the Role of Semantic
Overlapping as a Facilitator in Research Discoveries, In
Proceedings of the SDPS 2018 Workshop on
Accountability of AI, Bologna, Italy
Ayvaz, S., Horn, J. Hassanzadeh, O., Zhu, Q., Stan, J.,
Tatonetti, N. P., Vilar, S. Brochhausen, M., Samwald,
M. Rastegar-Mojarad, M. Dumontier, M., Boyce, N. P.
2015. Toward a complete dataset of drug–drug
interaction information from publicly available sources,
Journal of Biomedical Informatics 55 (2015) 206–217
Butte A. J., 2008. Translational bioinformatics: coming of
age. J Am Med Inform Assoc 15(6):709–714,
Available on-line, M2824v1
Doulaverakis, D., Nikolaidis G, Kleontas, A.,
Kompatsiaris, I. 2012. GalenOWL: Ontology-based
drug recommendations discovery. Journal of
Biomedical Semantics.2012 Dec 20; 3(1):14.
Grando, A., Farrish, S., Boyd, C., Boxwala, A., 2012.
Ontological approach for safe and effective
polypharmacy prescription. In AMIA, Annual
Symposium Proceedings 2012, 2012: pp. 291-300.
Herrero-Zazo, M., Segura-Bedmar, I., Hastings, J.,
Martínez, P. 2015. DINTO: Using OWL Ontologies
and SWRL Rules to Infer Drug-Drug Interactions and
their Mechanisms. Journal of Chemical Informatic
Modeing, 2015 Aug 24; 55(8):1698-707
Herrero-Zazo, M., Segura-Bedmar, I., Hastings, J.,
Martínez, P. Declerck 2013. The DDI corpus: An
annotated corpus with pharmacological substances and
drug–drug interactions. Journal of Biomedical
Informatics 46 (2013) 914–920
BIOINFORMATICS 2020 - 11th International Conference on Bioinformatics Models, Methods and Algorithms
274

Juric, R. (2016) Could Semantic Technologies Create a new
Computational Model Outside Semantic Web, in
Proceedings of the 21st SDPS 2016 Conference,
December 2016, FL, US.
Juric, R., Kim, I. (2017) Software Architectures for Smart
Applications which Merge Ontological Reasoning With
Big Data Analytics, In Proceedings of the 22nd SDPS
2017 conference November 2017
Juric, R., Almami, E. (2019) Proposing Drug Repositioning
though Semantic Reasoning, in Proceedings of the
22nd SDPS 2017 conference, July 2017, Taiwan.
Juric R. (2019) Semantic matching between Eligible
Paitents and clinical Trials using SWRL enabled OWL
Ontologies. In Proceedings of the 24th SDPS 2019
Conference, July 2019, Taiwan.
Kilicoglu H, Shin D, Fiszman M, Rosemblat G, Rindflesch
TC. SemMedDB: a PubMed-scale repository of
biomedical semantic predications. Bioinformatics
2012; 28:3158–60.
Kim, S., Liu, H., Yeganova, L., Wilbur, W. J. 2015.
Extracting drug–drug interactions from literature using
a rich feature-based linear kernel approach. Journal of
Biomedical Informatics 55 (2015) 23–30
Knox, C.; et al. DrugBank 3.0: a comprehensive resource
for “omics” research on drugs. Nucleic Acids Res.
2011, 39, D1035−41.
Liu, G. Wang, G. Chen (2019) Identifying Adverse Drug
Events from Social Media using an Improved Semi-
Supervised Method, in IEEE Intelligent Systems
January 2019 PP(99):1-1
Machado, C. M.; Rebholz-Schuhmann, D.; Freitas, A. T.;
Couto, F. M. The semantic web in translational
medicine: current applications and future directions.
Briefings Bioinf. 2015, 16, 89.
OWL/SWR. 2004. Ontology Web Language
https://www.w3.org/TR/owl-features/ and Semantic
Web Rule Language, available atT
https://www.w3.org/Submission/SWRL/
Patadia, R.; Kataria, P., Juric, R., Kim, I. “Conceptual
Design Of Semantic Software Applications” in
Proceedings of the 16th Intern. SDPS 2011 Conference,
Jeju Island, South Korea, June 12-16.
Payne Translational Informatics: Realizing the Promise
of Knowledge-Driven Healthcare, Payne, Philip R.O.,
Embi, Peter J. (Eds.) Springer-Verlag London 2015.
Segura-Bedmar, P. Martínez, C. de Pablo-Sánchez (2010)
A linguistic rule-based approach to extract drug-drug
interactions from pharmacological documents, From 4
th
Int. Workshop on Data and Text Mining in Biomedical
Informatics (DTMBio) 2010, Toronto, Canada.
October 2010
Shojanoori, R. 2013. Towards Formalization of Situation-
Specific Computations In Pervasive Computing
Environments”, PhD Thesis, University of
Westminster, London, UK, 2013.
Saaidi, R., Kataria, P., Juric R. 2011. Semantic
Management of Submissions of Applications for
Marketing Authorisation of Medicines. Book chapter,
in: Suh, S.C., Tanik, M., Gurupur, V. P. (Eds.)
Biomedical Engineering: Healthcare Systems,
Technology and Techniques (Springer; 1st Edition (20
Aug 2011).
Sahu, S. K., Anand A. 2018 Drug-Drug Interaction
Extraction from Biomedical Text Using Long Short
Term Memory Network, Journal of Biomedical
Informatics, Volume 86, October 2018, Pages 15-24
Saleha, A., Saba, T., Hussain, S., Ahmed, M., Akram, S. K.
Khan, S. K., Anjum, A., Shah, M.A., Javaid, N. 2017.
An ontology-based approach for detecting drug abuse
epidemiology, Journal of Medical Imaging and Health
Informatics, 2017, 7(6), pp. 1324-1337.
Sara, A., Mahmoud, H., El-Shishtawy, T. 2018. A
methodology for building an integrated drug interaction
ontology. International Journal of Advancements in
Computing Technology (IJACT), 2018, 10(2), pp. 1-10.
SWT. 2004. Semantic Web Technology SWT Road Map,
at W3C website https://www.w3.org/2001/10/03-sww-
1/slide7-0.html
Tarabi, M., Juric, R (2018) Software Architectures For
Smart Applications In The Management Of Chronic
Diseases: A Case Study Of Reversibility Of Diabetes 2,
In Proceedings of the 51st HICSS Conference, HI, US,
January 2018
Tsafnat G., Lin F., Choong M.K. (2013) Translational
Biomedical Informatics. In: Dubitzky W.,
Wolkenhauer O., Cho KH., Yokota H. (eds)
Encyclopedia of Systems Biology. Springer, New
York, NY
UMLS Reference Manual, National library of Medicine
(last update September 2009) available at
https://www.ncbi.nlm.nih.gov/books/NBK9676/pdf/B
ookshelf_NBK9676.pdf)
Yoshikawa, S., Satou, and T. Konagaya, A. 2004. Drug
interaction ontology (DIO) for inferences of possible
drug-drug interactions, Stud. Health Technol Inform.
2004; 107(Pt 1):454-8.
Zhang, R., Cairelli, M. J., Fiszman,., Rosemblat, M.,
Kilicoglu, H., Rindflesch, T. C., Pakhomova, S. V. ,
Melton, S. V. 2013. Using semantic predications to
uncover drug–drug interactions in clinical data, Journal
of Biomedical Informatics 49 (2014) 134–147
Zhoua, D., Miaoa, L., He Y. 2018. Position-aware deep
multi-task learning for drug–drug interaction
Extraction, Artificial Intelligence in Medicine 87 (2018)
1–8.
Adding Value to Translational Informatics through the Semantic Management of Drug to Drug Interaction
275
