
Automatic Segmentation of the Cervical Region in Colposcopic
Images
Paloma Cepeda Andrade, Sesh Commuri
University of Nevada, Reno, 1664 N Virginia St, Reno, NV, U.S.A.
Keywords: Cervical Cancer, Colposcopy, Image Segmentation, LAB Color Space, Morphological Filtering, K-means.
Abstract: Cervical cancer is one of the most common cancers affecting women, especially in developing countries and
in resource constrained areas in the western world. While easily treatable if detected early, the lack of adequate
resources and skilled physicians make this disease difficult to detect and treat. In this paper, we propose a
vision-based approach anchored in machine-learning principles to detect and quantify lesions on the surface
of the cervix. Preliminary results indicate that the proposed method can segment images of the cervix and
successfully detect lesions other artifacts. The image normalization approach can also determine the locations
of lesions and their spread. Validation of this approach during clinical trials is being pursued as the first step
towards developing low-cost bioinformatics-based screening tools for early detection of cervical cancer.
1 INTRODUCTION
Cervical cancer is the fourth most frequent cancer in
women worldwide and represents 6.6% of all cancers
affecting women (WHO, 2019). Most cervical cancer
cases are caused by various strains of the Human
Papilloma Virus (HPV). There are currently vaccines
that protect against common cancer-causing types of
HPV and can reduce the risk of cervical cancer.
Cervical cancer is treatable if detected and
diagnosed early. Typical screening methods for
detecting cervical cancer currently include techniques
such as Papanicolau (Pap) smear, HPV typing, and
colposcopy. Cost-effective options, such as Visual
Inspection with Acetic Acid (VIA), and Visual
Inspection with Lugol Iodine (VILI) are
recommended as the best screening methods in
developing countries (Sankaranarayanan et al., 2003).
Cryotherapy or loop electrosurgical excision
procedure (LEEP) (Basu & Sankaranarayanan, 2017)
can provide effective and appropriate treatment for
most women who screen positive for precancerous
lesions, and “screen-and-treat” and “screen, diagnose
and treat” are both valuable approaches.
Detection of cervical precancerous lesions can be
done by examining the cervix, vulva, and vagina
through a colposcope. This procedure, called
colposcopy, is used to detect genital warts,
inflammation of the cervix (cervicitis), and
precancerous changes in the tissue of the cervix,
vagina, and vulva. During colposcopy, the vaginal
walls are held open using a speculum and the surface
of the cervix is illuminated using a light source. The
colposcope (shown in Figure 1) is then used to
visually examine the cervix and the adjacent tissues.
Figure 1: Left: Standard colposcope. Right: Portable
colposcope (Basu & Sankaranarayanan, 2017).
During cervical examination, the cervix is first
rinsed with saline solution and then stained with 3-
5% acetic acid (Sankaranarayanan et al., 2004). If the
cervical epithelium contains an abnormal load of
cellular proteins, acetic acid causes these proteins to
coagulate. As a result, the affected areas appear
opaque or white under visual inspection. The higher
the grade of neoplasia, the greater the density of
acetowhitening (AW) (Li & Poirson, 2006). There are
66
Andrade, P. and Commuri, S.
Automatic Segmentation of the Cervical Region in Colposcopic Images.
DOI: 10.5220/0010835200003123
In Proceedings of the 15th International Joint Conference on Biomedical Engineering Systems and Technologies (BIOSTEC 2022) - Volume 1: BIODEVICES, pages 66-73
ISBN: 978-989-758-552-4; ISSN: 2184-4305
Copyright
c
2022 by SCITEPRESS – Science and Technology Publications, Lda. All rights reserved

also other characteristics such as morphological
changes (for example, tissue shape, mosaic, and
punctuation vessels) that can also indicate
abnormalities (Li & Poirson, 2006). Such factors
usually require an expert to determine diagnosis.
Abnormal growth of cells on the surface of the
cervix if untreated, can potentially lead to cervical
cancer. Cervical Intraepithelial Neoplasia (CIN) or
Cervical Dysplasia, refers to the potentially
precancerous transformation of cells of the cervix.
Presence of dysplasia can be further investigated
through Pap smear and biopsy. If detected early, CIN
can be managed by treatment and full recovery is
possible (Castle, Stoler, Solomon, & Schiffman,
2007). However, it is difficult to quantify different
characteristics of these lesions unless the patient is
examined by a trained professional. Even then,
diagnosis can be highly subjective and may lead to
misdiagnosis or an unnecessary biopsy (Castle et al.,
2007; Kudva & Prasad, 2018).
While screening and treatment has been quite
effective in reducing the occurrence of cancer in the
developed world, the need for expensive equipment,
trained colposcopists, and clinical infrastructure has
limited its benefit in other parts of the world. In 2018,
around 570,000 women were diagnosed with cervical
cancer worldwide, and nearly 311,000 women died
from this treatable disease (WHO, 2019). There are
many contributing factors to this alarmingly high rate
of death. These include social stigma, lack of
awareness and access to care, poor staining technique
during colposcopy, poor magnification and resolution
of the available devices in the market, cumbersome
nature of colposcopes due to size and need for
electrical power source, and the need for multiple
visits to the medical clinics, which can be unpleasant
and time-consuming experiences for the patient
(WHO, 2019).
Therefore, there is a need for a low-cost and
portable tool for screening and detecting abnormal
changes in the cervix that may later develop into
invasive cancer. Further, the system must be built
with automated detection and diagnosis capabilities
to enable use by health workers in the field where
experienced doctors and colposcopists are not
available.
Detection of lesions from images of the cervix has
been attempted by several researchers. These
approaches utilize digital images of the cervix
obtained during colposcopic examination. Staining
the cervix with acetic acid exposes any lesions
present as white areas (acetowhite regions) under
illumination. While these AW regions can be
detected, these regions are often distorted by the
presence of glare from the light source, reflections
from the speculum, as well as portions of the
speculum and other artifacts that are captured in the
image. Automatic detection of AW regions requires
the removal of glare, reflections, and other artefacts
from the image, extraction of the region of the cervix
that is of interest and detection of the AW regions.
Further, it is advantageous to locate the lesion and
determine its spread as such information can be used
for targeted biopsy instead of the four-quadrant
biopsy procedure that is commonly used.
In this paper, we present our approach to detection
of precancerous lesions on the cervix. In this
approach we first detect glare and specular reflection
(SR) in the image. This is followed by image
segmentation to identify the cervical region of interest
(ROI). The ROI is then used to detect the presence of
AW regions that are indicative of lesions. The use of
this algorithm in detecting lesions is demonstrated
using two colposcopy datasets that are available in the
public domain (add references). The method and
results are discussed in detail in Sections 3 and 4.
2 LITERATURE REVIEW
2.1 Acquisition of Cervical Images
Automated detection of CIN has benefited from
studies over the past several decades where digital
images of the cervix were collected. Initial efforts
include the Guanacaste Project in Costa Rica between
1993 and 1994 to investigate the role of HPV in the
development of CIN (Bratti et al., 2004). In this
project, researchers used a fixed-focus camera to
obtain cervical images of over ten thousand women.
Archived digitized cervical images from this project
were later used by Hu and co-researchers to develop
a method based on deep learning algorithm for
automated visual evaluation of cervical images (Hu et
al., 2019).
The ASCUS/LSIL Triage Study for Cervical
Cancer (ALTS) was a clinical trial conducted in the
United States between 1996 and 2000. ASCUS stands
for Atypical Squamous Cells of Undetermined
Significance and LSIL for Low-Grade Squamous
Intraepithelial Lesions (Schiffman & Adrianza,
2000). The images collected have also been used in
several research efforts (Greenspan et al., 2009; Guo
et al., 2020) to automate detection of precancerous
lesions on the cervix. While these two studies resulted
in a wealth of digital images that spurred research into
automated analysis of these cervical images, the lack
of uniformity in the imaging process, varying light
Automatic Segmentation of the Cervical Region in Colposcopic Images
67

intensity and focal lengths of the cameras, and
varying sizes of the images, make the analysis of
these images extremely difficult.
2.2 Detection and Removal of Glare
and Specular Reflections (SR)
One of the limiting factors in automatic detection of
CIN is the presence of specular reflections in cervical
images. The presence of moisture on the surface of
the cervix causes reflection of the light source. These
reflections, termed as Specular Reflections (SR),
appear as bright white spots in the cervigrams and
make it difficult to distinguish between AW regions
and reflections.
As mentioned in the previous section, staining the
cervix with acetic acid can expose potential
precancerous lesions. Under normal circumstances,
the epithelium remains transparent after the
application of acetic acid, but when a precancerous
lesion is present, the epithelium becomes opaque, and
the reflection of light gives it a white color (Basu &
Sankaranarayanan, 2017). An example of this
occurrence is shown in Figure 2. The image on the
left is of a cervix before the application of acetic acid.
The image on the right is of the same cervix, after
application of acetic acid, with presence of
acetowhite (AW) features. It can also be seen that SR
on the cervical image will also appear white in color,
but with high brightness and low saturation values.
This presents a problem because it can blend in with
the AW features of interest and produce incorrect
classification or diagnostic results. Therefore, it is
necessary to identify these SR areas prior to any
medical assessment.
Figure 2: Cervix before (left) and after (right) application
of acetic acid. The AW area (blue line) is made visible after
applying acetic acid (Basu & Sankaranarayanan, 2017).
Several researchers have attempted to recover
information occluded by glare and SR. Lange (Lange,
2005) implemented adaptive thresholding to obtain
glare feature maps and remove glare, followed by a
filling algorithm to estimate the color and texture in
the affected areas. Meslouhi and co-investigators
(Meslouhi et al., 2011) proposed an automatic glare
extraction and feature inpainting method based on
obtaining and comparing luminance components
from both RGB and CIE-XYZ color spaces. Das and
Bhattarcharyya (Das et al., 2011) developed an
algorithm to perform glare removal through the
design of morphological filters and with subsequent
inpainting using inward interpolation from the pixels
outside the detected area. Bai and co-researchers (Bai
et al., 2018) also aim to detect areas with SR and
restore the area through a filling algorithm.
Region-filling algorithms may provide an
estimate of what the area would look like without SR.
However, this estimate may forego some texture
features that can be of importance in deciding the
severity of a precancerous lesion.
2.3 Extraction of the Region of Interest
Color images and the correct representation of
features are essential when working with digital
medical images. The most common representation of
color in images in computer monitors is the sRGB
(standard RGB) color space. RGB stands for the three
primary colors: red, green, and blue. Images
represented in this color space are formed by
combining arrays containing values of these
individual color components. For most digital
images, every possible color can be formed with
values in the range of (0, 255) for each primary color
component, in the form: 𝐶
𝑖
= (𝑅
𝑖
, 𝐺
𝑖
, 𝐵
𝑖
) where
0 ≤ 𝑅
𝑖
, 𝐺
𝑖
, 𝐵
𝑖
≤ 255 (Burger & Burge, 2016). While
sRGB is an accepted color standard for screens, it is
limited when trying to extract desired features, as
there are no light or saturation components, which are
good indicators of a lesion present on the cervix.
A different color representation of an sRGB
image can make it easier to extract meaningful
information. The LAB color space was developed to
linearize the representation with respect to human
color perception and to generate a more intuitive
color space. Color is expressed as three dimensions:
L* which represents luminosity; and a* and b*, which
are the components that specify hue and saturation
along a green-red and blue-yellow axis, respectively
(Burger & Burge, 2016). An implementation of this
color space to observe a cervical image is illustrated
in Figure 3. Note that the L* channel presents
information pertaining to the brightness in the image,
a useful feature in detecting AW regions.
Figure 3: Cervix image converted to CIELAB color space,
left to right: Original; L* channel; a*channel; b* channel.
BIODEVICES 2022 - 15th International Conference on Biomedical Electronics and Devices
68

Analysis of a cervical image can be difficult due
to the presence of artifacts such as the speculum in the
image. These artifacts do not present useful
information and are detrimental to the automated
detection of lesions. Therefore, detection of the
cervical area plays an important role in assessment of
colposcopic images by highlighting only the region of
interest (ROI). Accurate detection of lesions on a
cervix involves removal of glare and SR on the
cervical epithelium, accurate delineation of the
cervical region, and identification, location, and
spread of lesions represented by AW regions.
Figure 4 shows a color image of a cervix. This
image contains the speculum used in colposcopy, as
well as interference by the reflection of light on the
cervical epithelium. Manual delineation of the region
of interest and the AW region diagnosed as Cervical
Intraepithelial Neoplasia of Grade 1 (CIN1) are also
presented.
Figure 4: Manual segmentation of the cervical region of
interest (ROI), acetowhite staining (AW), and specular
reflection (SR).
Several researchers have investigated automatic
segmentation of the cervical region to detect lesions.
Using hue, saturation, and brightness (HSV color
space) information in images, Bai and co-researchers
were able to segment cervical images and extract the
ROI. Their approach was shown to have accuracy,
specificity, and sensitivity of 87.25%, 81.99% and
96.70%, respectively (Bai et al., 2018) Das and
Choudhury (Das & Choudhury, 2017) also attempted
to obtain the cervical ROI and detect lesions by
transforming cervical images into the HSV color
space. However, they do not provide validation
information to demonstrate that their method can
reliably extract the ROI in digital images of the
cervix.
Tariq and Burney (Tariq & Burney, 2014) used k-
means segmentation to extract the ROI from images
of the cervix. In their analysis, they found that k-
means ROI segmentation results in better accuracy
when images in LAB color space are used in
comparison to RGB images. Das and other
investigators (Das et al., 2011) proposed an algorithm
to detect the cervical ROI and AW lesions using the
L* channel of the LAB color space. They achieved
accuracy between 89% and 91% but provide no other
quantitative metrics to validate their approach.
While these results demonstrate the advantages of
using image analysis techniques to study cervigrams,
existing results in literature do not adequately address
all the challenges in analyzing images of the cervix.
3 METHODS
In this paper, we present a five-step approach to
analyzing cervigrams and detecting the presence of
lesions on the cervix. These steps are as follows:
1. Convert the cervical image from sRGB to
LAB color space and combine the
information from the L* and a* channels.
2. Use the k-means algorithm to obtain clusters,
segment the image, and identify the cervical
ROI.
3. Implement morphological filters to eliminate
holes and connect similar regions.
4. Automatically crop the segmented and
filtered image to maximize the ROI.
5. Identify AW lesions and calculate their area
in proportion to the cervix.
These steps will be further discussed in the next
subsections.
3.1 Data Acquisition
We have developed a prototype of the Cervitude
Imaging System (CIS). This prototype consists of a
probe equipped with a ranging sensor, a camera, and
LEDs for illuminating the cervix. The device includes
a microprocessor circuit that can control the intensity
of the LEDs and provides two-way communication
with the CIS application running on a host system
such as a computer, Android/iPhone, or a tablet. The
CIS application is built around an image analysis
algorithm that can quickly detect and locate lesions
on the surface of the cervix. With this device, we aim
to provide a portable, low-cost, solution to
communities that lack the necessary resources and
medical personnel. We are currently in the process of
performing a clinical study to perform preliminary
testing and to obtain our images using this prototype.
Two small datasets of cervical images that are
available online were used to demonstrate the
algorithms presented in this paper.
The first dataset used was obtained through the
Atlas of Colposcopy (Basu & Sankaranarayanan).
The Atlas of Colposcopy was developed by the
International Agency for Research on Cancer (IARC)
Automatic Segmentation of the Cervical Region in Colposcopic Images
69

and provides detailed information on everything
related to colposcopy. It is designed to serve as a
comprehensive manual for anyone working in this
area, both in the medical field and academic research.
This website contains a small repository of images
(107 in total) labeled in order of severity, from
“Normal” to “CIN-1”, “CIN-2”, “CIN-3”, and
“Carcinoma in situ (CIS)”. These images are of
relatively high quality, with a size of 600 x 800 pixels,
which makes them suitable for the segmentation
approach presented in this paper. The main drawback
is that there are no annotations available regarding the
cervical ROI, so a qualitative evaluation on the
segmentation results will be made.
The next image dataset that was used for analysis
is publicly available online
1
by Fernandes and co-
researchers (Fernandes et al., 2017). This dataset
contains a total of 284 colposcopic images with
annotations for the cervical ROI. Out of these, we will
use 91 images, which represent the cervical region
after acetic acid has been used. The annotations
provided will be used to validate our segmentation
algorithm. However, a downside is that there is no
pathological information available, so AW lesion
segmentation cannot be validated with this dataset.
3.2 Image Pre-processing
Our image pre-processing approach addresses the
challenges in cervical image analysis in the following
ways: by identifying regions containing SR, and by
preparing the image for k-means segmentation
through conversion of the images to LAB color space.
From Figure 3, it can be observed that both the L* and
a* channels can provide meaningful information.
Therefore, this method uses the information obtained
after combining these two channels, as they will bring
out the areas that present a high level of brightness
and absence of pink or red colors. The results of
combining these two channels are shown on Figure 5.
Figure 5: Visualization of the combination of L* and a*
channels. Left: original images. Right: resulting images.
1
https://archive.ics.uci.edu/ml/datasets
The results from Figure 5 show a way to interpret
specular reflections as an undesired feature for
diagnosing a precancerous lesion. Image (a) is that of
a healthy cervix, so correctly detecting these natural
features is essential to avoid over-treating a patient
for a misdiagnosed illness. Similarly, image (b)
shows the correct identification of areas with SR,
while leaving out the AW region.
3.3 Segmentation of the Cervical
Region
Image segmentation is a critical step when processing
images for classification. Through this process, an
image can be split into a given number of regions. The
k-means algorithm is a type of unsupervised
clustering algorithm commonly used for image
segmentation (Luo et al., 2003) due to its simplicity
and speed. It divides a dataset (in this case, an image)
into K classes, such that similar regions are clustered
together. The objective is to find the center of each
cluster C
i
and to assign all sample points in the
vicinity of the center to that cluster (Tariq & Burney,
2014).
The center of each cluster is the mean of the data
points which belong to it. A Euclidean distance
measurement:
𝐷𝑥, 𝑦
‖
𝑥𝑦
‖
(1)
is used to determine which cluster a data point
belongs to. The algorithm works as follows:
1. For a given number of K, the data points are
randomly grouped into each cluster.
2. The center of each cluster is calculated.
3. The distance from each point to each cluster
center is calculated.
4. Each point is reassigned to the nearest
cluster.
Steps 2-4 are repeated iteratively until there are no
changes in the grouping.
The input to our k-means segmentation algorithm
is the image resulting from the element-wise
multiplication of the L* and a* channels. Then, we
implement the k-means algorithm to obtain three
clusters. Finally, we implement morphological
filtering using a hole-filling operation, followed by an
opening operation, which removes small dark spots
on the image, and connects the bigger, white, areas.
In most cases, this will leave our resulting binary
mask, which will indicate the cervical ROI. In a few
BIODEVICES 2022 - 15th International Conference on Biomedical Electronics and Devices
70
cases, two or more resulting areas will appear. If that
occurs, the largest area will be considered as the ROI.
4 RESULTS AND DISCUSSION
4.1 Atlas of Colposcopy Dataset
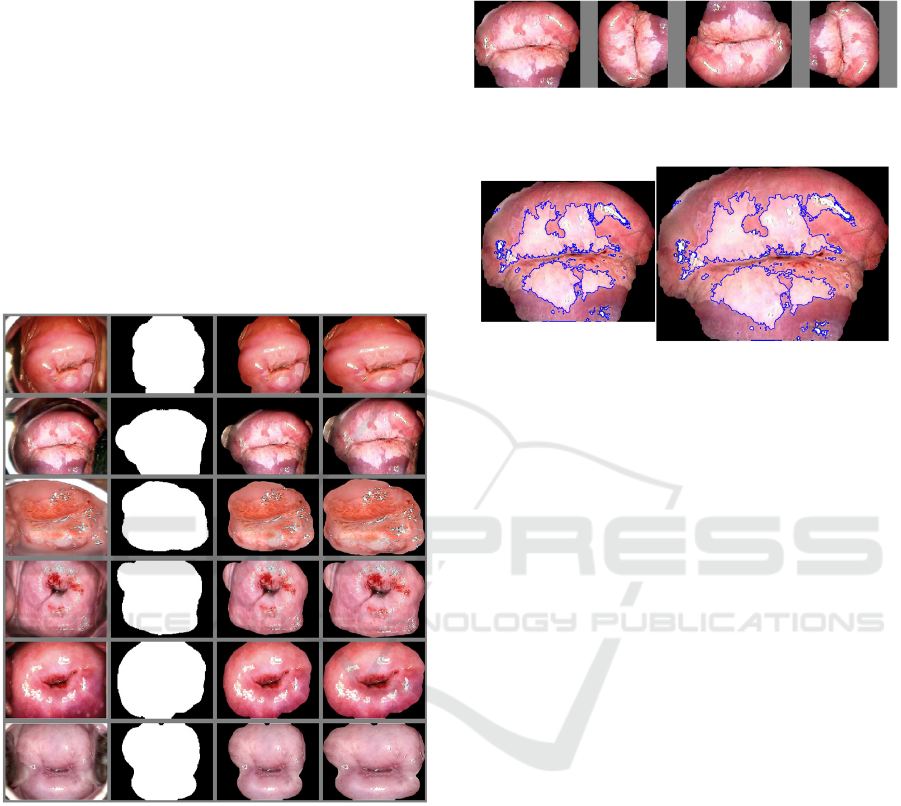
Figure 6 shows the segmentation results on the Atlas
of Colposcopy dataset. The first column shows the
original RGB colposcopy image. The second column
displays the resulting outline after LAB color space
conversion and our segmentation algorithm. The third
column represents the segmented colposcopic image,
without the speculum and outer edges of the cervical
region.
Figure 6: ROI Segmentation Results. Left to right: Original
image; binary mask; segmented image; cropped and resized
segmented image.
Additionally, an auto-crop function was created to
ensure that the cervical area remains at the center of
the processed image. The results are shown on the
fourth column in Figure 6. If the image is rotated, the
region of interest will remain at the center. An
example is illustrated in Figure 7. The size of the
segmented and cropped image is 486 x 599 pixels.
To detect the presence of AW lesions, the k-
means algorithm was used to perform automatic
segmentation of the image in Figure 7. The image
represents a cervix with CIN 2, where a portion of the
cervix is covered by AW lesions. Without the
presence of the speculum and the background, the
AW region detection was successful. The result is
shown on Figure 8.
Figure 7: Segmentation and auto-cropping of cervical
image after 90-degree rotation.
Figure 8: AW lesion detection results. Left: Cropped image.
Right: Cropped and resized image.
From Figure 8, the area of the identified AW
regions in the cropped image covers 35.39% of the
cervical ROI. When the image is resized to its original
dimensions, the segmentation algorithm detects that
the AW area covers 35.34% of the cervical ROI. This
shows that when the cropped and segmented image is
resized to the original dimensions, the AW area
proportion remains within algorithmic precision.
This shows that the ROI segmentation and auto-
cropping will retain the same ratio of AW region with
respect to the area of the cervix. The process is almost
unaffected by the size and orientation of the original
image. Therefore, the procedure can be used during
cervical screening using portable colposcopes or
other imaging devices. Further, the procedure affords
a simple way to compare screening results from
several patient visits and can be used to document the
efficacy of the treatment regimen.
4.2 Fernandes Dataset
We also performed our ROI segmentation algorithm
on the images from the Fernandes dataset. Figure 9
shows our results compared to the provided
annotations. The first column contains the original
images, the second column shows our segmentation
results with true negative values in black, true
positive values in white, false negative values in blue,
and false positive values in red. The third column
shows the ground truth annotations, and the fourth
column shows the final, segmented image.
Automatic Segmentation of the Cervical Region in Colposcopic Images
71
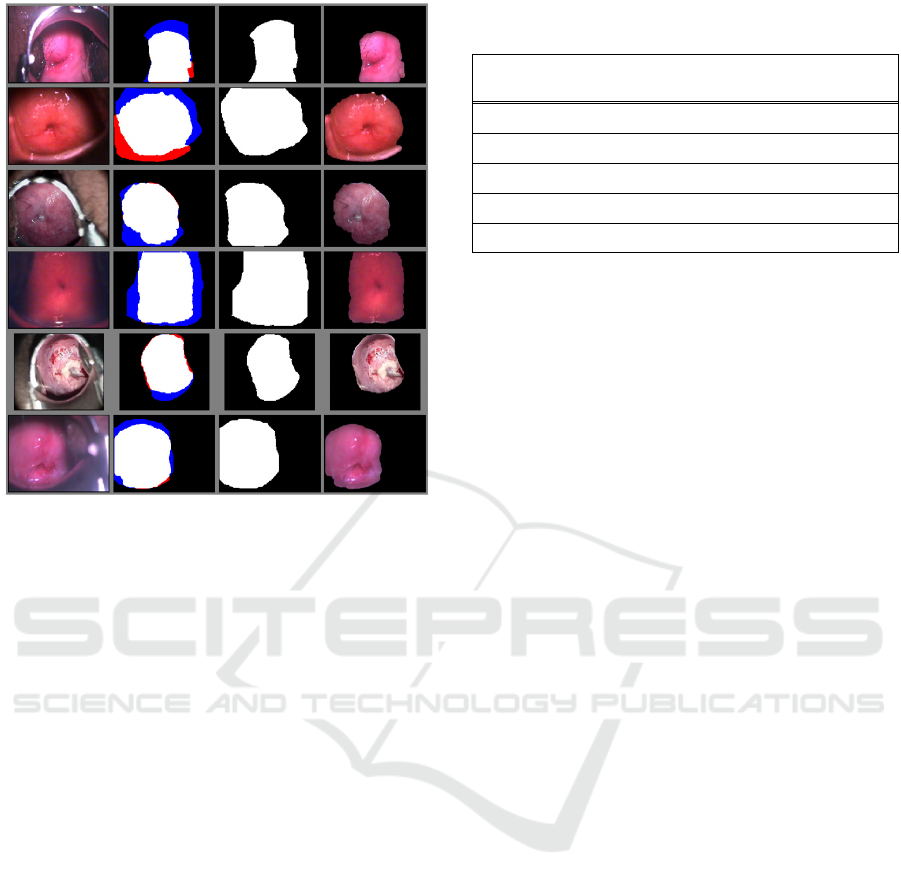
Figure 9: ROI Segmentation Results. Left to right: Original
image; binary mask with false negative segmentation in
blue, and false positive segmentation in red; ground truth;
segmented image.
The false negative (FN) values are those which are
annotated as cervix, and which our algorithm
classified as non-cervix. While we consider this
dataset to be of relatively good quality, in many cases
the illumination in the images is not adequate, and
shadows present in them can affect the performance
of our algorithm. We observed that most of our errors
are FN, where our algorithm did not include
shadowed areas in the cervix as part of the ROI. Even
though we do not have annotations in our other
dataset, a qualitative assessment of our results
indicates that if the ROI is well illuminated, our FN
rate will go down.
The quantitative evaluation of our algorithm is
shown in Table 1. Our results show that the average
accuracy of the segmentation algorithm was 83.33%.
In fact, 67 out of 91 images were segmented with
accuracy over 80%. Specificity indicates the
proportion at which our algorithm can classify pixels
that are not part of the ROI. The average specificity
was almost 87%. Our average sensitivity is about
81%, which shows the rate at which we can correctly
identify a pixel as part of the ROI. Precision is the
measurement indicating the possibility that the pixel
classified as part of the cervix is not part of the
background (Gerig et al., 2001). The average
precision is 80.53%.
Table 1: Accuracy and performance of our segmentation
algorithm.
Performance
Metric
Max
(%)
Min
(%)
Median
(%)
Mean
(%)
F1 Score 95.50 47.36 82.07 79.46
Accuracy 97.44 58.32 83.84 83.33
Specificity 100 58.77 88.83 86.63
Sensitivity 99.72 33.24 81.58 81.12
Precision 100 42.82 81.73 80.53
Our experimental results show that our
segmentation algorithm performs in a suitable
manner, with accuracy, specificity, and sensitivity of
up to 97.44%, 100%, and 99.72%, respectively. This
means that the algorithm can segment the cervical
region in clinical practice with excellent results. We
have shown that, given a small amount of data, an
algorithm can be developed to identify the cervical
ROI, regardless of position or distance from the
colposcope to the cervix. Further improvements are
expected as we complete our clinical study.
5 CONCLUSIONS
In this paper, an image analysis-based approach for
screening for cervical cancer was presented.
Preliminary results indicate that the proposed method
can segment images of the cervix, reduce the effect of
glare from light sources, remove specular reflections
and other artifacts, and successfully detect lesions.
While the method was validated using sample images
from the Atlas of Colposcopy and the Fernandes
dataset, extensive analysis must be conducted using a
variety of images collected in the field to improve the
sensitivity and specificity of the method in obtaining
the cervical ROI and detecting cervical dysplasia.
Validation of this approach during clinical trials is
being pursued by the authors as the first step towards
developing low-cost bioinformatics-based screening
tools for early detection of cervical cancer. Accurate
automatic detection of cervical dysplasia can prove to
be crucial in regions where medical experts or clinical
resources are not available.
REFERENCES
(WHO), W. H. O. (2019). Cervical Cancer.
https://www.who.int/cancer/prevention/diagnosis-
screening/cervical-cancer/en/
BIODEVICES 2022 - 15th International Conference on Biomedical Electronics and Devices
72

Bai, B., Liu, P. Z., Du, Y. Z., & Luo, Y. M. (2018).
Automatic segmentation of cervical region in
colposcopic images using K-means. Australas Phys
Eng Sci Med, 41(4), 1077-1085. https://doi.org/
10.1007/s13246-018-0678-z
Basu, P., & Sankaranarayanan, R. (2017). Atlas of
Colposcopy – Principles and Practice. IARC
CancerBase. https://screening.iarc.fr/atlascolpo.php
Bratti, M. C., Rodríguez, A. C., Schiffman, M., Hildesheim,
A., Morales, J., Alfaro, M., Herrero, R. (2004).
Description of a seven-year prospective study of human
papillomavirus infection and cervical neoplasia among
10000 women in Guanacaste, Costa Rica, Rev Panam
Salud Publica, 15(2), 75-89. https://doi.org/10.1590/
s1020-49892004000200002
Burger, W., & Burge, M. J. (2016). Digital Image
Processing (2 ed.). Springer-Verlag London.
https://doi.org/10.1007/978-1-4471-6684-9
Castle, P. E., Stoler, M. H., Solomon, D., & Schiffman, M.
(2007). The relationship of community biopsy-
diagnosed cervical intraepithelial neoplasia grade 2 to
the quality control pathology-reviewed diagnoses: an
ALTS report. Am J Clin Pathol, 127(5), 805-815.
https://doi.org/10.1309/PT3PNC1QL2F4D2VL
Das, A., Avijit, K., & Bhattacharyya, D. (2011).
Elimination of specular reflection and identification of
ROI: The first step in automated detection of Cervical
Cancer using Digital Colposcopy 2011 IEEE
International Conference on Imaging Systems and
Techniques.
Das, A., & Choudhury, A. (2017). A novel humanitarian
technology for early detection of cervical neoplasia:
ROI extraction and SR detection. 2017 IEEE Region 10
Humanitarian Technology Conference (R10-HTC),
Dhaka.
Fernandes, K., Cardoso, J. S., & Fernandes, J. (2017).
Transfer Learning with Partial Observability Applied to
Cervical Cancer Screening. In L. A. Alexandre, J.
Salvador Sánchez, & J. M. F. Rodrigues, Pattern
Recognition and Image Analysis Cham.
Gerig, G., Jomier, M., & Chakos, M. (2001). Valmet: A
New Validation Tool for Assessing and Improving 3D
Object Segmentation. In W. J. Niessen & M. A.
Viergever, Medical Image Computing and Computer-
Assisted Intervention – MICCAI 2001 Berlin,
Heidelberg.
Greenspan, H., Gordon, S., Zimmerman, G., Lotenberg, S.,
Jeronimo, J., Antani, S., & Long, R. (2009). Automatic
detection of anatomical landmarks in uterine cervix
images. IEEE Trans Med Imaging, 28(3), 454-468.
https://doi.org/10.1109/TMI.2008.2007823
Guo, P., Xue, Z., Long, L. R., & Antani, S. (2020). Cross-
Dataset Evaluation of Deep Learning Networks for
Uterine Cervix Segmentation. Diagnostics (Basel),
10(1).
Hu, L., Bell, D., Antani, S., Xue, Z., Yu, K., Horning, M.
P., Schiffman, M. (2019). An Observational Study of
Deep Learning and Automated Evaluation of Cervical
Images for Cancer Screening. J Natl Cancer Inst
,
111(9), 923-932. https://doi.org/10.1093/jnci/djy225
Kudva, V., & Prasad, K. (2018). Pattern Classification of
Images from Acetic Acid-Based Cervical Cancer
Screening: A Review. Crit Rev Biomed Eng, 46(2),
117-133. https://doi.org/10.1615/CritRevBiomedEng.2
018026017
Lange, H. (2005). Automatic glare removal in reflectance
imagery of the uterine cervix. Proceedings of SPIE -
The International Society for Optical Engineering,
Li, W., & Poirson, A. (2006). Detection and
Characterization of Abnormal Vascular Patterns in
Automated Cervical Image Analysis. In G. Bebis, R.
Boyle, B. Parvin, D. Koracin, P. Remagnino, A. Nefian,
G. Meenakshisundaram, V. Pascucci, J. Zara, J.
Molineros, H. Theisel, & T. Malzbender, Advances in
Visual Computing Berlin, Heidelberg.
Luo, M., Ma, Y.-F., & Zhang, H.-J. (2003). A Spatial
Constrained K-means A pproach to Image
Segmentation Joint Conference of the Fourth
International Conference on Information,
Communications and Signal Processing, 2003 and
Fourth Pacific Rim Conference on Multimedia
Singapore.
Meslouhi, O., Kardouchi, M., Allali, H., Gadi, T., &
Benkaddour, Y. (2011). Automatic detection and
inpainting of specular reflections for colposcopic
images. Open Computer Science, 1(3).
Sankaranarayanan, R., Shastri, S. S., Basu, P., Mahé, C.,
Mandal, R., Amin, G., Dinshaw, K. (2004). The role of
low-level magnification in visual inspection with acetic
acid for the early detection of cervical neoplasia.
Cancer Detect Prev, 28(5), 345-351. https://doi.org/
10.1016/j.cdp.2004.04.004
Sankaranarayanan, R., Wesley, R., Thara, S., Dhakad, N.,
Chandralekha, B., Sebastian, P., Nair, M. K. (2003).
Test characteristics of visual inspection with 4% acetic
acid (VIA) and Lugol's iodine (VILI) in cervical cancer
screening in Kerala, India. Int J Cancer, 106(3), 404-
408. https://doi.org/10.1002/ijc.11245
Schiffman, M., & Adrianza, M. E. (2000). ASCUS-LSIL
Triage Study. Design, methods and characteristics of
trial participants. Acta Cytol, 44(5), 726-742.
https://doi.org/10.1159/000328554
Tariq, H., & Burney, S. M. A. (2014). K-Means Cluster
Analysis for Image Segmentation. International
Journal of Computer Applications, 96.
Automatic Segmentation of the Cervical Region in Colposcopic Images
73
