
Production of Biodiesel from Microalgae Using Transesterification
Batch Reactor with the Assistance of Calcium Oxide Hydrotalcite
Catalyst
Yustia Wulandari Mirzayanti
a
, M. Amirul Fatah
b
, Rendy Meydianto A. Y. Tengker
c
,
and Dian Yanuarita Purwaningsih
d
Department of Chemichal Engineering, Institute Technology Adhi Tama Surabaya,
Arif Rahman Hakim No. 100, Surabaya, Indonesia
Keywords: Alternative Energy, Biodiesel, Microalga Nannochloropsis sp., In-Situ Batch Reactor, Transesterification,
Cao/Hydrotalcite.
Abstract: Research the process of making biodiesel as alternative energy using microalgae Nannochloropsis s.p using
the In-Situ transesterification method. The manufacturing process is carried out in two stages: in situ
transesterification using a batch reactor and distillation process purification. The research was carried out
using the fixed variable, namely the molar ratio of oil, algae content: solvent, namely 1:20, and the
independent variable stirring speed in batch reactors of 50, 100, 150, 200, and 250 rpm with the catalyst used
being a CaO/Hydrotalcite heterogeneous catalyst. This study obtained the highest crude biodiesel yield of
38.36% and the lowest Free Fatty Acid (FFA) content of 2.76 mg NaOH/gram sample at a stirring speed of
250 rpm.
1 INTRODUCTION
The Energy and Mineral Resources (EMR) sector is a
sector that plays a vital role in supporting the national
economy. The Energy and Mineral Resources sector
includes coal, new and renewable energy, oil and gas.
Currently, Indonesia still has a high dependence on
fossil energy, where dependence on coal is 38%, oil
and natural gas is 51%, while on new and renewable
energy, it is still relatively low at 11%. Since 2004,
Indonesia has become an oil importer country. In
2020, energy consumption increased due to the
COVID-19 pandemic, where production is
approximately 700 thousand bpd, and consumption is
1.5 million bpd. Biodiesel is a biofuel that is an
alternative to petroleum diesel fuel. Biodiesel is an
ester hydrocarbon compound derived from long-
chain fatty acids. The components of biodiesel
depend on the type of material used as raw material
a
https://orcid.org/0000-0001-8647-9638
b
https://orcid.org/0009-0003-7550-5820
c
https://orcid.org/0009-0005-3249-7562
d
https://orcid.org/0000-0002-0554-5005
because it is related to the chemical structure of oil or
lipid compounds produced from raw materials, such
as the number of carbon atoms, as well as the number
of double bonds in the lipid hydrocarbon bonds. The
main raw material for making biodiesel in Indonesia
today is Crude Palm Oil (CPO). At the same time,
Crude Palm Oil is also the raw material for making
cooking oil, so if biodiesel production expands, it can
cause a potential scarcity of cooking oil. The cause of
biodiesel is currently an alternative fuel that is quite
popular because biodiesel is an environmentally
friendly fuel, its raw materials are easy to get, and the
manufacturing process is not complex. Biodiesel is
generally made from vegetable materials, including
plant oils and animal fats.
Microalgae can absorb solar energy and bind CO
2
efficiently, producing energy through photosynthesis
(Astuti and Sriwuryandari, 2010). Microalgae can be
cultivated easily because they use solar energy to
180
Mirzayanti, Y., Fatah, M., Tengker, R. and Purwaningsih, D.
Production of Biodiesel from Microalgae Using Transesterification Batch Reactor with the Assistance of Calcium Oxide Hydrotalcite Catalyst.
DOI: 10.5220/0012107900003680
In Proceedings of the 4th International Conference on Advanced Engineering and Technology (ICATECH 2023), pages 180-185
ISBN: 978-989-758-663-7; ISSN: 2975-948X
Copyright
c
2023 by SCITEPRESS – Science and Technology Publications, Lda. Under CC license (CC BY-NC-ND 4.0)

photosynthesize. The high growth rate allows
microalgae to produce more biodiesel feedstock than
plant oils or animal fats. Some microalgae have a high
lipid content which can be used as an alternative in
biodiesel feedstock. The total lipid content of each
microalga varies from 20-50%, but some microalgae
have a lipid content of more than 40% (Patmawati et
al, 2014). The use of microalgae as an alternative raw
material for biodiesel production has considerable
potential. Indonesia occupies the third position in
replacing fossil fuels with microalgae biodiesel in the
future (Batten et al, 2011). The reason microalgae is
a raw material that has considerable potential is the
characteristics of the microalgae itself. Microalgae is
biomass with a high growth rate, easy to cultivate,
environmentally friendly, easy to dry, and has a high
lipid content that is easy to extract.
In a previous study, Edo et al. (2020) researched
making biodiesel from the microalgae
Nannochloropsis sp. using the in-situ
transesterification method with the help of a sulfuric
acid catalyst. The results showed that the variable
molar ratio of microalgae to methanol 1:7 showed the
highest crude biodiesel yield of 12% and the lowest
FFA content of 0.399 mg NaOH/gr (Mirzayanti et al,
2020). In the same year, Septianto et al. (2020) also
researched making biodiesel from the microalgae
Nannochloropsis sp. using the transesterification
method with the help of a CaO/hydrotalcite catalyst.
The results showed that at a molar ratio of oil to
methanol 1:15 with 10% catalyst weight%, the
highest Fatty Acid Methyl Ester (FAME) content was
61.77%, and biodiesel yield was 60.39% (Septianto et
al, 2020). In another research conducted by Retya et
al. (2022) on the production of biodiesel from the
microalgae Nannochloropsis sp. using the
microwave-assisted in situ transesterification method
with the help of a NaOH catalyst, the results showed
a molar ratio of oil to methanol of 1:10 with a catalyst
concentration of 0.4 M NaOH to obtain a biodiesel
yield of 60.39% and an FFA of 0.0088 mg NaOH/gr
(Retya et al, 2022).
In this research, the Nannochloropsis sp.
microalgae type is used as a raw material for biodiesel
using the In-Situ transesterification method. The
catalyst used was a CaO/hydrotalcite heterogeneous
catalyst with methanol as a solvent as well as a
reactant in the in-situ transesterification process and
n-hexane as a co-solvent which was carried out using
a batch reactor that had been designed. The research
used a fixed variable, the molar ratio of oil algae
content and solvent 1:20, and the independent
variable is stirrer speed in a batch reactor.
2 LITERATURE REVIEW
Microalgae are classified as autotrophic organisms
that reproduce through the process of photosynthesis.
The unicellular structure found in microalgae allows
them to convert solar energy into chemical energy
easily (Liu et al, 2016). It is estimated that microalgae
can produce 200 times more oil content than other oil-
producing plants (Sharma et al, 2012). Microalgae are
also known as marine plants with advantages over
other fuel sources. They do not require large areas,
produce biomass quickly, and use CO
2
gas in the
growth process to reduce air pollution (Gultom,
2018). Generally, there are three different microalgae
growth conditions: phototrophic, heterotrophic, and
mixed. Under phototrophic conditions, microalgae
are highly dependent on sunlight as energy and CO
2
as carbon sources. Phototrophic conditions are often
referred to as autotrophic photosynthesis. The second
condition, namely the growth of heterotrophic
microalgae, requires organic carbon as an energy
source. Some commonly used organic nutrient
sources include glucose, acetate, and glycerol.
Microalgae growth is also influenced by its
physiological properties (Hindarso et al, 2015). The
physiology of this microalgae species can affect
nutrient uptake and its culture media. Naturally,
microalgae plants that live in open ponds will quickly
grow to cover the pond's surface. If the microalgae
physiology is very good, these conditions can
stimulate biomass production with high oil and starch
content. However, certain physiological responses do
not allow microalgae to survive in these ponds. Of the
several microalgae studied, the type of microalgae
Nannochloropsis sp. can produce lipid products.
Lipid content in microalgae Nannochloropsis sp.
is quite high, namely 31% to 68% of its dry weight
(Chisti, 2013). Nannochloropsis sp. is microalgae
that are yellow-green, round, small in size, and 2-4
µm in diameter (Salam et al, 2016). There are
membrane-enclosed cell walls, mitochondria,
chloroplasts, and nuclei in the morphology of
Nannochloropsis sp. The chloroplast is bell-shaped,
located at the edge of the cell, and has a light-sensitive
stigma. Nannochloropsis sp. has six species:
Nannochloropsis graditana, Nannochloropsis
granulata, Nannochloropsis limnetic,
Nannochloropsis oceanica, Nannochloropsis salina,
Nannochloropsis oculate. Microalgae also have an
essential function in marine ecosystems. Acts as a
natural food for zooplankton and fish larvae because
it is rich in carbohydrates, proteins, fats, minerals, and
amino acids. Nannochloropsis sp. is a widely
cultivated microalga and is rich in benefits, especially
Production of Biodiesel from Microalgae Using Transesterification Batch Reactor with the Assistance of Calcium Oxide Hydrotalcite
Catalyst
181

regarding health. It is perfect for consumption by
zooplankton, such as rotifers, because it has a high
content of Eicosapentaenoic Acid (EPA) and
Docosahexaenoic Acid (DHA) (Meria et al, 2021).
The transesterification reaction is a chemical
reaction between triglycerides and alcohol that uses a
catalyst to produce monomers known as biodiesel
products. Common solvents in the method other than
methanol are NaOH, carbonates, and alkoxides such
as sodium methoxide, CO
2
, sodium propoxide, and
sodium butoxide. Several kinds of catalysts can be
used in the transesterification method, such as an
acid, alkaline, or heterogeneous catalyst. In the
transesterification method, a reaction occurs to form
triglycerides contained in the raw material with
methanol as a solvent using a catalyst. During the
reaction process, triglycerides create methyl esters,
fatty acids, and glycerol; the glycerol layer will
decrease in the existing biodiesel. In addition, there is
an in-situ transesterification method, a direct
transesterification process of biomass rich in lipids or
oil without knowing the extract and the purification
process separately. During the process, the raw
material will directly react with alcohol using a
catalyst. This transesterification process can coincide
with the response or first reactant process followed by
an extraction process (Panjaitan et al, 2018).
3 EXPERIMENTAL METHODS
3.1 Preparation of Catalyst
Catalyst CaO/hydrotalcite was prepared by the
method of incipient wetness impregnation.
Hydrotalcite as much as 10 grams in the oven for 12
hours at a temperature of 100
o
C. CaCO
3
is calcined to
CaO in a furnace for 3 hours at 900°C then weighed
with a loading ratio of 1:1 to hydrotalcite. After the
CaO is calcined, the CaO is cooled and dissolved in
distilled water. The incipient wetness impregnation
method was used for hydrotalcite by spraying it with
CaO solution in stages. The CaO-Hydrotalcite
mixture dried in an oven for 12 hours at 100°C. The
dry CaO-Hydrotalcite mix was then calcined in a
furnace for 3 hours at 900°C (Heraldy et al, 2017).
3.2 In Situ Transesterification
Microalgae Nannochloropsis sp. was weighed and
then put into a batch reactor along with methanol and
CaO/hydrotalcite catalyst according to the variables
set. The top nozzle of the reactor is closed, and the
stirrer and reactor heater are turned on with the
stirring speed according to the variable set until the
reaction time has been determined. After the reaction
time has been reached, the product mixture is
removed from the reactor through the bottom nozzle
and then cooled to room temperature. The residue in
the product mixture is separated from the filtrate
using a filtrate vacuum. The filtered residue was
washed using 30 mL of a combination of methanol-
n-hexane 1:1 (v/v) to recover the remaining biofuel.
The filtrate was put into a separatory funnel, n-hexane
was added with a ratio of 1:1 (v/v), then shaken and
allowed to stand until two layers were formed for 30
minutes. The bottom layer was removed from the
separatory funnel and added n-hexane with a ratio of
1:1 (v/v), then shaken and allowed to stand until two
layers were formed for 8 hours. The top layer is
washed using distilled water to bind the remaining
catalyst and glycerol, which is still left in the mixture.
The top layer is considered a transesterification
product and then refined to separate the biofuel and
solvent.
3.3 Refining
The product from the transesterification reaction is
put into a boiling flask, then placed on a hotplate, and
a solvent container is placed at the end of the
condenser. The heater is turned on until the
temperature in the boiling flask reaches 70 °C.
Heating can be stopped if all the n-hexane solvent has
evaporated with no more steam dripping at the end of
the condenser. The distillation product is cooled
down to room temperature. The distillate product was
put into the oven at 70 °C for 4 hours to evaporate the
solvent and water contained in the product. The
product is cooled and then analyzed for its yield and
acid number (SNI 7182:2015). The formula for yield
calculation is as follows:
Yield =
× 100%
(1)
The formula for Free Fatty Acid (FFA) calculation is
as follows:
FFA =
× ×
(2)
MW
N
V
W
= Molecular mass
= Concentration of NaOH
= Consumption of NaOH
= Wei
g
ht of crude biodiesel
ICATECH 2023 - International Conference on Advanced Engineering and Technology
182
4 RESULTS AND DISCUSSION
Research on making biodiesel from microalgae
Nannochloropsis sp. using the in-situ
transesterification batch reactor with the help of a
CaO/hydrotalcite catalyst was carried out in 3 stages,
namely the catalyst synthesis stage, the
transesterification stage, and the distillation stage.
The catalyst synthesis stage is carried out to
manufacture CaO/hydrotalcite catalyst using the
incipient wetness impregnation method, where
Hydrotalcite is impregnated by spraying it with CaO
solution according to the specified variables. CaO is
obtained from the compound CaCO
3,
calcined at 900
°C. In a study by Royani et al. (2016), when CaCO
3
is calcined to a temperature of 900 °C, it will
decompose into CaO and CO
2
(Royani et al, 2016). In
the In-Situ transesterification stage, a series of
processes run simultaneously. The first is extracting
oil from the base material, which is carried out by
methanol and co-solvent n-hexane. Then the
triglycerides react with methanol to form methyl
esters in the transesterification reaction. Methanol is
a reactant in the transesterification reaction and a
lipid-extracting solution contained in microalgae. N-
hexane acts as a co-solvent in the extraction process.
Adding n-hexane as a co-solvent can increase the
lipid extracted from microalgae (Panjaitan et al,
2018). CaO/Hydrotalcite catalyst can speed up the
transesterification reaction at the in-situ
transesterification stage (Januar, 2014).
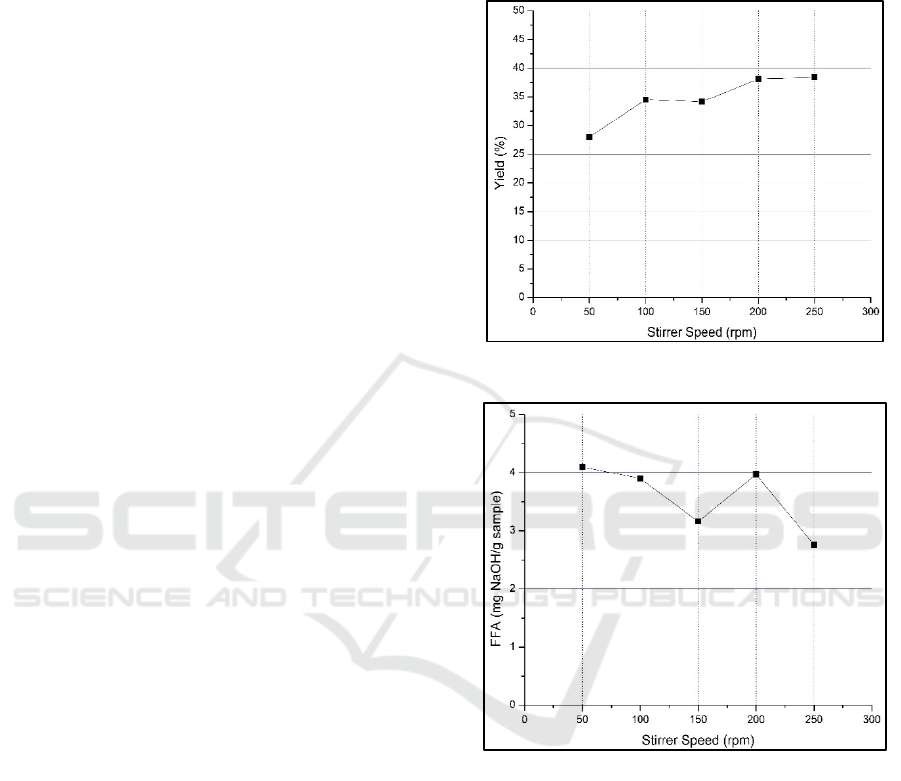
Figure 1 shows the effect of stirring speed on
biodiesel yield based on variations in reactor stirring
speed. Figure 1 shows that the stirring speed affects
the yield of crude products formed. The faster the
stirring speed is given, the more yield of crude
products is formed. It happens because the stirring
speed can increase the material particles' movement.
Collision activity and contact between one particle
and another material particle are becoming more
frequent, causing chemical reactions to occur more
rapidly (Miskah et al, 2017). Adding n-hexane to
transesterification can optimize the yield result
because n-hexane as a co-solvent can contact first
with the lipid in microalgae and then reacts with
methanol (Dianursanti, 2015). The highest yield of
crude product is found at the variable stirring speed
of 250 rpm, producing a crude yield of 38.36%. The
results obtained were lower when compared to the
previous study by Retya (2022), which was 71.7%.
This is because the method used is in situ microwave-
assisted transesterifications. Microwave radiation is
more effective in breaking down microalgae cells
(Barqi, 2015; Khan, 2021). Therefore, the lipid
extraction process runs faster. Methanol, as a solvent,
is also very good at absorbing microwave radiation to
support the transesterification reaction (Retya et al,
2022).
Figure 1: Effect of Stirrer Speed on Yield Biodiesel
Figure 2: Effect of Stirrer Speed on Free Fatty Acid Content
of Biodiesel
Figure 2 shows the effect of stirring speed on FFA
crude levels based on stirrer speed variations. Figure
2 shows that the stirring rate affects the formed FFA
crude product. The FFA crude product that is formed
is related to the yield of the produced crude product.
The greater the yield of crude product, the smaller the
FFA crude product formed. It happens because the
conversion of biodiesel becomes larger and
suppresses the conversion of FFA formation. FFA
levels in crude biodiesel products must be minimized
because FFA can react with metals at high
temperatures inside the engine when biodiesel is used
(Adhani et al, 2016). In the FFA test results, the
lowest FFA content was obtained at the variable
Production of Biodiesel from Microalgae Using Transesterification Batch Reactor with the Assistance of Calcium Oxide Hydrotalcite
Catalyst
183

stirring speed of 250 rpm with an acid number content
of 2.76 mg NaOH/gram sample. The acid value in
biodiesel is still above the Standar Nasional
Indonesia (SNI) 7182: 2015 quality standard, which
is 0.5 mg NaOH/gram sample. The FFA level in the
biodiesel product formed is still higher when
compared to research conducted by Baqi (2022), and
Julrohiniar (2022), where the FFA levels produced
were 0.52 mg NaOH/gram sample and 0.321 mg
NaOH/gram sample. It is due to the biodiesel
manufactured by Baqi (2022) using a base catalyst
KOH and Julrohiniar (2022) using an SO
4
2-
/TiO
2
base
catalyst in which the contained fatty acids are
transformed into dimethyl ether (Baqi et al, 2022;
Julrohiniar et al, 2022). The crude biodiesel product
is shown in Figure 3.
Figure 3: The Crude Biodiesel Product
5 CONCLUSION
Based on this research, it can be concluded that
biodiesel production from microalgae
Nannochloropsis sp with CaO/hydrotalcite catalyst
using the in-situ transesterification batch reactor
produced the highest yield is 38.36% at a reactor
stirring speed of 250 rpm, and the lowest Free Fatty
Acid content is 2.76 mg NaOH/gram of sample at a
reactor stirring speed of 250 rpm.
REFERENCES
Adhani, L., Aziz, I., Nurbayti, S. and Octavia, C. A.,
(2016). Pembuatan biodiesel dengan cara adsorpsi dan
transesterifikasi dari minyak goreng bekas. Kimia
Valensi Journal, vol. 2, page 71-80.
Astuti, J. T., Sriwuryandari, L. (2010). Biodiesel dari
mikroalga: perbanyakan biomassa melalui penambahan
nutrisi secara bertahap. Bionatura Journal, vol. 12,
page 160-168.
Baqi, F., Putri, S. I., and Mirzayanti, Y. W. (2022). Proses
pembuatan biodiesel dari mikroalga nannochloropsis
sp. menggunakan metode transesterifikasi in-situ
dengan katalis KOH. Equilibrium Chemical
Engineering Journal. vol. 6, page 92-97.
Barqi, W. S. (2015). Pengambilan minyak mikroalga
chlorella sp. dengan metode microwave assisted
extraction. JBAT Journal, vol. 4, page 34-41.
Batten, D. F., Campbell, P. K., and Threlfall, G. (2011).
Resource potential of algae for sustainable biodiesel
production in the APEC economies. Australia: Asia-
Pacific Economic Cooperation.
Chisti, Y. (2013). Constraint to commercialization of algal
fuel. Biotechnolology Advanced Journal, vol. 167, page
201-214.
Dianursanti, Religia, P., and Wijanarko, A. (2015).
Utilization of n-hexane as co-solvent to increase
biodiesel yield on direct transesterification reaction
from marine microalgae. Procedia Environmental
Sciences 23, International Conferences on Tropical and
Coastal Region Eco-Development, page 412-420.
Gultom, S. O. (2018). Mikroalga: sumber energi terbarukan
masa depan. Marine Science Technolology Journal,
vol. 11, page 95-103.
Heraldy, E., Nugrahaningtyas, K. D., and Heriyanto.
(2017). Calcination on ca-mg-al hydrotalcite from brine
water and its characterization. ALCHEMY Journal, vol.
13, page 205-216.
Hindarso, H., Aylianawati, and Sianto, M. (2015).
Biodiesel production from the microalgae
nannochloropsis by microwave using CaO and MgO
catalysts. International Journal of Renewable Energy
Development, vol. 4, page 71-76.
Januar, I. (2014). Pengaruh kalsinasihidrotalsit
terhadapaktivitas katalitik pada reaksi transesterifikasi
etil asetat menjadi metil asetat. Indonesian Journal of
Chemical Research, page 26-37.
Julrohiniar, A., Ayuni, S. P. and Mirzayanti, Y. W. (2022).
Pembuatan biodiesel dari mikroalga nannochloropsis
sp. menggunakan metode transesterifikasi in-situ
dengan katalis SO
4
2-
/TiO
2
. Seminar Nasional Teknologi
Industri Berkelanjutan II (SENASTITAN II) Journal,
vol. 2, page 476-480.
Khan, H., Iqbal, T., Mujtaba, M. A., Soudagar, M. E. M.,
Veza, I., and Fattah, I. M. R. (2021). Microwave-
assisted biodiesel production using heterogeneous
catalysts. Energies Journal, vol. 14, page 1-16.
Liu, J., Sun, Z., and Gerken, H. (2016). Recent advances in
microalgal biotechnology. Foster City: OMICS Group.
Meria, R., Puspitasari. W., and Zulfahmi, I. (2021). Teknik
kultur nannochloropsis sp. skala laboratoriumdi balai
perikanan budidaya air payauujung bate. Banda Aceh:
Balai Budidaya Air Payau Ujung Batee.
Mirzayanti, Y. W., Syafutra, R. E., Vinata, Y., and Irawan,
C. (2020). Pembuatan biodiesel dari mikroalga
nannochloropsis sp. menggunakan metode
transesterifikasi insitu dengan bantuan katalis asam
sulfat. Buletin Profesi Insinyur Journal, vol. 4, page 80-
84.
Miskah, S., Apriani, R. and Miranda, D., (2017). Pengaruh
waktu reaksi dan kecepatan pengadukan terhadap
konversi biodiesel dari lemak ayam dengan proses
ICATECH 2023 - International Conference on Advanced Engineering and Technology
184

transesterifikasi. Teknik Kimia Journal, vol. 23, page
57-66.
Panjaitan, R. M., Mujaddid, F., Kalsum, U., and Qadariyah,
L. (2018). Biodiesel production from dry microalga
biomass by microwave-assisted in-situ
transesterification. The 24
th
Regional Symposium on
Chemical Engineering, MATEC Web of Conferences,
vol. 156, article no. 06005.
Patmawati, Ibrahim, B., Setyaningsih, I., and Sudadi, U.
(2014). Produksi biodiesel dari biomassa
chlamydomonas sp. ICBB 9113 dikultivasi
menggunakan media yang murah: efektifitas dari
beberapa metode ekstraksi. Widyariset Journal, vol.
17, page 269-276.
Retya, A. H. M., Cahyani, A. S. N., and Mirzayanti, Y. W.
(2022). Pembuatan biodiesel mikroalga
nannochloropsis sp. menggunakan metode in-situ
transesterification microwave-assisted dengan katalis
sodium hidroksida. Seminar Nasional Teknologi
Industri Berkelanjutan II (SENASTITAN II) Journal,
vol. 2, page 234-241.
Royani, A., Sulistiyono, E., and Sufiandi, D., (2016).
Pengaruh suhu kalsinasi pada proses dekomposisi
dolomit. Sains Materi Indonesia Journal, vol. 18, page
41-46.
Salam, K., Velasquez-orta, S., and Harvey, A. (2016).
Surfactant-assisted direct biodiesel production from
wet Nannochloropsis occulata by in situ
transesterification/reactive extraction. Biofuel Research
Journal, vol. 3, page 366-371.
Septianto, A. D., Aji, S., and Mirzayanti, Y. W. (2020).
Produksi biodiesel dari mikroalga nannochloropsis sp.
menggunakan metode transesterifikasi dengan bantuan
katalis heterogen CaO/hydrotalcite. Seminar Nasional
Sains dan Teknologi Terapan VIII Journal, vol. 1, page
493-498.
Sharma, K., Schuhmann, H., and Schenk, P. (2012). High
lipid induction in microalgae for biodiesel production.
Energies Journal, vol. 5, page 1532-1553.
Production of Biodiesel from Microalgae Using Transesterification Batch Reactor with the Assistance of Calcium Oxide Hydrotalcite
Catalyst
185
