
Diffusion Model for Generating Synthetic Contrast Enhanced CT
from Non-Enhanced Heart Axial CT Images
Victor Rogerio Sousa Ferreira
1
, Anselmo Cardoso de Paiva
1
, Aristofanes Correa Silva
1
,
João Dallyson Sousa de Almeida
1
, Geraldo Braz Junior
1
and Francesco Renna
2
1
Universidade Federal do Maranhão, Av. dos Portugueses, 1966, Bacanga,
São Luís/MA, Núcleo de computação Aplicada, UFMA, São Luís, Brazil
2
INESC TEC, Faculdade de Ciências da Universidade do Porto, Porto, Portugal
Keywords: Diffusion Models, Image Translation, Adversarial Networks.
Abstract: This work proposes the use of a deep learning-based adversarial diffusion model to address the translation of
contrast-enhanced from non-contrast-enhanced computed tomography (CT) images of the heart. The study
overcomes challenges in medical image translation by combining concepts from generative adversarial
networks (GANs) and diffusion models. Results were evaluated using the Peak signal to noise ratio (PSNR)
and structural index similarity (SSIM) to demonstrate the model's effectiveness in generating contrast images
while preserving quality and visual similarity. Despite successes, Root Mean Square Error (RMSE) analysis
indicates persistent challenges, highlighting the need for continuous improvements. The intersection of GANs
and diffusion models promises future advancements, significantly contributing to clinical practice. The table
compares CyTran, CycleGAN, and Pix2Pix networks with the proposed model, indicating directions for
improvement.
1 INTRODUCTION
Non-communicable diseases (NCDs) are medical
conditions that cannot be spread directly from one
person to another and are often caused by a
confluence of behavioural, physiological,
environmental, and genetic variables.
In (WHO, 2023), it is stated that NCDs are the world's
greatest cause of mortality, accounting for 41 million
deaths per year (74% of all deaths worldwide). The
bulk of NCD-related fatalities (17.9 million/year) are
attributable to cardiovascular diseases, which also
have a significant role in premature death and
disability globally (Dondi, 2021).
A timely and efficient way to improve population
health overall is through diagnostic imaging. By early
discovery, they can be utilised as a preventive
approach to lessen cardiovascular issues.
A common imaging modality for diagnosing
cardiovascular disorders is computed tomography
(CT) (Corballis, 2023; Counselor, 2023)—an
imaging modality with growing diagnostic utility.
Cardiac CT plays a crucial role in diagnosing and
managing heart diseases. It is possible to obtain
detailed three-dimensional images of the heart
through cardiac CT, allowing for precise evaluation
of cardiac anatomy, function, and circulation. This
makes cardiac CT a valuable tool for diagnosing
various heart conditions, including coronary artery
disease, cardiomyopathies, and congenital heart
defects
Cardiac CT is particularly useful in detecting
coronary artery disease (CAD), one of the leading
causes of morbidity and mortality worldwide.
Coronary artery disease (CAD) is a significant
cardiovascular disease defined by a narrowing or
blockage of the coronary arteries. With cardiac CT,
the existence and severity of CAD may be evaluated
non-invasively. Coronary calcium deposits, linked to
an elevated risk of coronary artery disease, are often
evaluated by non-contrast cardiac computed
tomography (NCCT). On the other hand, contrast-
enhanced cardiac computed tomography (CECT) is
used when the goal is to quantify cardiovascular
disease, evaluate blood flow dynamics, define the
composition of plaque, and offer quantitative
measurements of disease severity. This tomography
technique injects contrast materials into the body to
increase the contrast between certain organs, blood
Ferreira, V., Cardoso de Paiva, A., Silva, A., Sousa de Almeida, J., Braz Junior, G. and Renna, F.
Diffusion Model for Generating Synthetic Contrast Enhanced CT from Non-Enhanced Heart Axial CT Images.
DOI: 10.5220/0012724600003690
Paper published under CC license (CC BY-NC-ND 4.0)
In Proceedings of the 26th International Conference on Enter prise Information Systems (ICEIS 2024) - Volume 1, pages 857-864
ISBN: 978-989-758-692-7; ISSN: 2184-4992
Proceedings Copyright © 2024 by SCITEPRESS – Science and Technology Publications, Lda.
857

arteries, or tissues and the surrounding structures on
CT images.
CECT improves patient outcomes by helping
physicians detect and track many elements of
cardiovascular disease. It does this by making
cardiovascular structures and abnormalities more
visible.
But in contrast to NECT, CECT is more costly,
involves more radiation exposure, and may have
unfavourable side effects, including headaches and
vomiting. Furthermore, anyone with allergies or renal
problems might be in danger when using CECT.
There has been great potential for using artificial
intelligence in cardiac CT to improve diagnosis and
prognosis. This exam has unique characteristics that
make it even more attractive for the application of
artificial intelligence, although the complexity of this
application is increasing.
Another application is in the assessment of
cardiac function from cardiac CT images. Algorithms
can automatically analyse cardiac volumes, ejection
fraction, and wall motion, providing precise
measurements that aid in evaluating heart function.
This automated analysis saves time and resources,
allowing physicians to focus on more complex
interpretations and personalised treatment plans for
each patient.
Among these challenges, we highlight the need
for quantitative evaluations, which generally involve
quantitative evaluations such as ventricular volume,
fraction of blood volume ejected out of the ventricle,
volumetric evaluation of heart muscle tissue, amount
of plaque present in the coronary arteries, area of
stenosis, among others. In addition, the images are
acquired with thinner slices, and the evaluation
targets are smaller (e.g., coronary arteries).
Given the potential risks associated with CECT,
generative AI-based techniques for cardiac imaging
can assist professionals in assessing coronary artery
disease without the drawbacks of CECT. More
precisely, we can create a CECT image that matches
the given NECT image using data-driven methods
without requiring contrast substance injection.
The challenge of medical image synthesis may be
approached through picture-to-image translation
(Parmar, 2023) or style transfer (Jing, 2020). This
topic poses extra complications in the context of
cardiac CT images. Since the same patient's NECT
and CECT images are frequently significantly out of
alignment, direct monitoring for NECT to CECT
mapping is rarely feasible.
In recent years, medical image translation has
emerged as a powerful solution to overcome these
challenges. This process involves synthesising
images of the target modality based on the guidance
of images acquired from the source modality.
However, the inherent nonlinear variations in tissue
signals between modalities make this problem
complex and ill-conditioned.
Learning-based methods, especially Generative
Adversarial Networks (GANs), have shown
remarkable success in image translation tasks. GANs
employ an adversarial mechanism in which a
discriminator guides a generator to perform a one-
time mapping to produce the target image. While
GANs exhibit exceptional realism in image synthesis,
they indirectly characterize the target modality
distribution, potentially introducing biases and
limiting the mapping process's reliability.
As an alternative approach, recent studies in
computer vision have explored diffusion models
based on explicit likelihood characterisation and a
gradual sampling process to enhance sample fidelity.
However, the potential of diffusion methods in
medical image translation remains largely
unexplored, partly due to computational challenges
and difficulties in the non-paired training of regular
diffusion models.
In this work, we propose a deep learning-enabled
image-to-image translation model that can map
contrast-free CT images of the heart to contrast-
enhanced ones. To achieve this, we implemented an
adversarial diffusion model, applying concepts from
GANs to generate high-quality images. This method
aims to provide an accurate model compared to other
approaches.
2 RELATED WORKS
In (Azarfar, 2023), authors present several papers
proposing deep learning architectures to reduce or
eliminate administered contrast media to acquire
clinically useful computer tomographies.
The introduction of GANs (Goodfellow, 2014)
presented an innovative approach to generative
models. GANs operate based on the principle of
rivalry between two networks - the generator and the
discriminator. The generator aims to produce
synthetic data indistinguishable from real data, while
the discriminator strives to differentiate between the
two. Through adversarial training, GANs achieve
Nash equilibrium, converging the generator's
distribution to the training data.
In image translation, especially in the analysis of
medical images, GANs are widely used for their
ability to automatically learn patterns in input data so
that the model can generate new examples (output)
ICEIS 2024 - 26th International Conference on Enterprise Information Systems
858

that could exist in the original dataset. When
performing image generation, the simplest model
maps from source to destination through a trained
generator using adversarial loss (Goodfellow, 2014).
Consequently, the GAN-based translation approach
has been extensively adopted in various applications.
Conditional GANs excel in mapping a single
source to a destination, improving sensitivity to high-
frequency details in tissue structure compared to
traditional pixel-to-pixel losses. Integrating
adversarial loss terms has proven effective in
enhancing spatial accuracy and realism in target
images synthesised with GANs, surpassing
conventional convolutional models.
Other studies, specifically using GANs, whose
primary focus lies in the synthesis of contrast-
enhanced computed tomography (CECT) images
from non-contrast CT (NCT) scans, are (Chun, 2022)
and (Seo, 2021). They employ a two-stage framework
and sophisticated network architectures as generators,
such as DenseNet and SPADE (Park, 2019). They
successfully align NCT and CECT images,
surpassing previous methods in accuracy and
applicability.
Other research extends to artery-contrasted
computed tomography (ACT), which is crucial for
diagnosing conditions like aneurysms. To mitigate
the risks of contrast agents, they introduce an aorta-
aware deep learning approach that synthesises artery-
contrasted CT volumes directly from non-contrast CT
data (Hu, 2022). Utilising aGANs and innovative loss
functions, their model demonstrates remarkable
accuracy in estimating ACT slices, thus enhancing
diagnostic precision while minimising patient risk.
The Pix2Pix (Zhu, 2017) model is one of the
approaches designed for image-to-image translation
tasks. It consists of a generator to create synthetic
images and a discriminator to distinguish between
real and generated images. Training involves an
adversarial process, where the generator tries to
deceive the discriminator, and the discriminator seeks
to identify fake images.
Applications of the Pix2Pix architecture (Choi,
2021) utilise the fundamental structure of the original
pix2pix model to generate synthetic contrast
enhanced from non-contrast chest CT, with the
distinction that the 2D convolutional layers are
substituted by their 3D equivalents. This model
comprises a generator and discriminator networks
akin to a conventional GAN. The generator network
is a U-Net convolutional neural network encoder-
decoder with skip connections. The discriminator
network is a PatchGAN that classifies each pixel
patch as real or fake, and its convolutional module is
identical to the encoder block of the generator.
A dissertation (Domingues, 2022) compares the
performance of two GAN models, Pix2Pix-GAN and
Cycle-GAN, in generating contrast-enhanced images
from non-contrast CT scans. The study explores the
trade-offs of using 2D, 2.5D, and 3D inputs,
employing different types of generators and datasets.
Evaluation metrics include Structural Similarity
Index Measure (SSIM), Peak Signal-to-Noise Ratio
(PSNR), Mean-Square Error (MSE), and Dice metric
for high contrast region fidelity.
CyTran (Ristea, 2021) is a GAN-based model
designed for working with CT images. This
innovative approach focuses on bidirectional
translation of contrast and non-contrast computed
tomography (CT) scans, even when the images lack
direct pairing. CyTran aims to address the challenge
of generating contrast scans for patients who cannot
receive contrast and to enhance the alignment
between contrast and non-contrast CT scans. The
method employs a cycle-consistent architecture based
on generative adversarial transformers designed for
transferring CT scans across different contrast
phases. Inspired by the CycleGAN framework,
CyTran comprises two discriminators and two
generators, enabling training on unpaired images
through a multi-level cycle consistency loss. In
addition to ensuring image-level consistency, Cytran
utilises additional losses between intermediate
feature representations to enhance the model's
performance further. This comprehensive strategy
contributes to the model's effectiveness in translating
CT images bi-directionally, offering valuable
applications in medical imaging scenarios.
However, GANs present their challenges. Issues
such as lower reliability in mapping a single sample,
premature convergence of the discriminator, and poor
representational diversity leading to mode collapse
can compromise the quality and diversity of
generated samples. Despite these challenges, GANs
currently lead in image generation tasks, surpassing
other models based on metrics such as Inception
Score and Accuracy.
Lately, deep diffusion models have become an
alternative to GANs in generative modelling tasks in
computer vision (Yang, 2022). These models are
inspired by non-equilibrium thermodynamics,
defining a Markov chain of diffusion steps to add
random noise to the data slowly and then learning to
reverse the diffusion process to construct desired data
samples from the noise. Noise removal is conducted
by a neural network architecture trained to maximise
the correlation between adjacent pixels.
Diffusion Model for Generating Synthetic Contrast Enhanced CT from Non-Enhanced Heart Axial CT Images
859
This diffusion technique provides greater
reliability in network mapping and improves the
quality and diversity of generated samples. The two-
step structured diffusion model starts with direct
diffusion, where input data is gradually perturbed
over multiple steps by adding Gaussian noise. In the
reverse step, the model is trained to recover the
original data, reversing the diffusion process step by
step. This innovative method offers a robust and
effective approach to generating realistic data in
various computer vision contexts.
Moreover, diffusion models are easily adaptable,
able to use different architectures such as
Transformers (Peebles, 2023) and adversarial
networks (Wang, 2023), achieving results that
surpass the quality of previous diffusion models in
metrics like peak signal-to-noise ratio (PSNR), ratio
is used as a quality measurement between the original
and a compressed image, and structural similarity
index measure (SSIM) for measuring the similarity.
Table 1: Overview of the applied techniques cited in related
works.
Related Works Applied Techniques
(Ristea, 2021) CycleGan structure with
Pix2Pix + Transformers
(Seo, 2021) GAN
(
SPADE +DCGAN
)
(Chun, 2022) GAN
(FC-DenseNet + PatchGAN)
(
Choi, 2021
)
Pix2Pix
(Domingues, 2022) CycleGan and Pix2Pix with
SkipResidual Generato
r
(
Park, 2019
)
SPADE
3 METHODOLOGIES
This work proposes a methodology for synthesising
NECT to CECT images, utilising a GAN-based
approach with diffusion models. This methodology
consists of the following steps: data acquisition, data
pre-processing and proposed network architecture.
3.1 Data Acquisition
The dataset was obtained at the Orca Score in the
Grand Challenge platform (Wolterink,2022). Images
in this dataset were acquired on four different CT
scanners from four different vendors in four different
hospitals using standard parameters for calcium
scoring in cardiac CT. For each patient, both a non-
contrast-enhanced CT and a contrast-enhanced
computed tomography angiography (CTA) image are
provided. The training set consists of images of 32
patients. The test set consists of images of 40 patients.
From this dataset, 6209 images were extracted,
divided into 2812 for testing and 3397 for training and
validation. The entire set consists of images with and
without contrast from the same patients.
3.2 Data Pre-Processing
For this study, it was essential to conduct specific
preprocessing steps before utilising these CT images
to enhance the overall quality of the model. The
preprocessing involved segregating the slices of
contrast and non-contrast CT images of each patient's
heart, selecting those in the same position with a high
similarity index. This approach ensured that only the
most relevant and corresponding images were used to
refine the model's analysis.
To achieve this, the images were correlated using
the SSIM and Normalised Cross-Correlation (NCC)
to assess the structural similarity.
Images with higher similarity indices were
subsequently considered equivalent. Following this,
the best images from each patient, meaning those with
the same position and the highest similarity indices
correlating contrast and non-contrast, were separated
and allocated into training, testing, and validation
sets. The number of retained images was as follows.
Table 2: Orca Dataset Paired Filtration Summary.
Contrast Non-Contrast
Train 200 200
Test 100 100
Validation 50 50
3.3 Proposed Network Architecture
Based on the SynDiff network (Özbey, 2023), a
diffusion model was developed with a conditional
origin adversarial projector for fast and accurate
reverse diffusion sampling. Unlike conventional
models that use a relatively large number of steps, this
network employs fast-forward diffusion, adaptively
adjusting noise variance to balance efficiency and
precision in image generation.
The proposed network utilises a Cycle-GAN
architecture consisting of diffusive generators and a
non-diffusive discriminator (Figure 1). The diffuse
ICEIS 2024 - 26th International Conference on Enterprise Information Systems
860
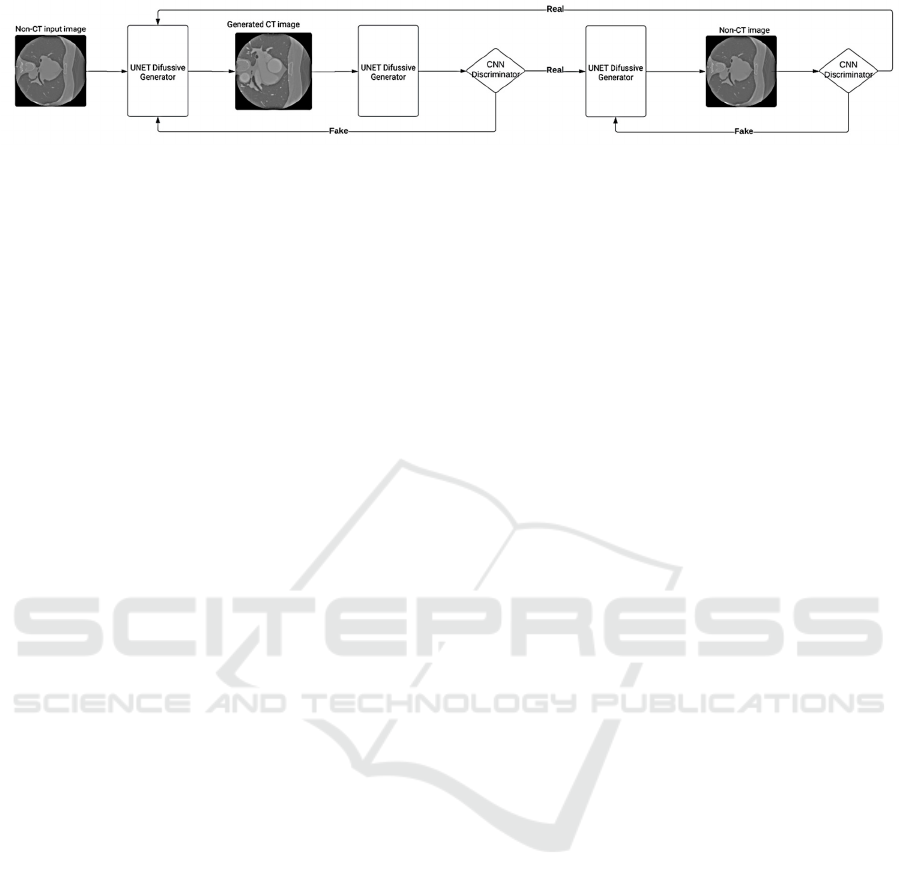
Figure 1: Architecture of proposed network.
generator translates images from the NECT to the
CECT domain and vice versa. Conversely,
discriminators aim to distinguish between real and
generated images. In the diffusive module, generators
employ a UNet backbone comprising six encoding
and decoding blocks (Ho, 2020). Each block includes
two residual subblocks followed by a convolutional
layer. During encoding, the convolutional layer
reduces the feature map resolution by half, while the
channel dimensionality is doubled every other block.
The convolutional layer doubles the resolution for
decoding, while the channel dimensionality is halved
every other block.
The proposed network utilises a Cycle-GAN
architecture consisting of diffusive generators and a
non-diffusive discriminator (Figure 1). The diffuse
generator translates images from the NECT to the
CECT domain and vice versa. Conversely,
discriminators aim to distinguish between real and
generated images. In the diffusive module, generators
employ a UNet backbone comprising six encoding
and decoding blocks (Ho, 2020). Each block includes
two residual subblocks followed by a convolutional
layer. During encoding, the convolutional layer
reduces the feature map resolution by half, while the
channel dimensionality is doubled every other block.
The convolutional layer doubles the resolution for
decoding, while the channel dimensionality is halved
every other block.
The discriminator model is designed as a
sequential neural network (Radford, 2015), tailored
for input images of size 256 by 256 pixels. It consists
of two convolutional layers with 64 and 128 filters,
each with a kernel size of (5, 5) and a stride of (2, 2)
for downsampling. Leaky ReLU activation functions
introduce non-linearity after each convolutional
layer. To prevent overfitting, dropout layers with a
dropout rate of 0.3 are incorporated after each Leaky
ReLU layer.
Given the larger input dimensions, the
architecture is adapted to handle the increased spatial
information. Following the convolutional layers, a
flattening layer transforms the 2D feature maps into a
1D vector. Finally, a dense layer with one neuron is
added, serving as the output layer for binary
classification (discriminating between real and
generated CT images).
During training, the proposed network enforces
cycle consistency, a crucial property that ensures the
translated images maintain semantic content and
realism. By incorporating an additional loss function
to quantify the disparity between the NECT image
generated by the second generator and the original
NECT image, as well as vice versa, the proposed
network promotes cycle consistency. This
regularisation technique guides the generator models
in the creation of CECT images. The generators aim
to minimise both the adversarial loss, which measures
their ability to generate realistic images, and the
cycle-consistency loss simultaneously. Meanwhile,
discriminators are trained to improve their ability to
distinguish between real and generated CT images,
thereby providing feedback to the generators.
The training objective of the proposed network
resembles the CycleGAN method, which utilises two
main loss functions: adversarial loss and cycle-
consistency loss. Adversarial loss incentivises the
generators to produce images indistinguishable from
real images, as perceived by the discriminators. On
the other hand, cycle-consistency loss enforces the
constraint that translating an image from one domain
to another and then back should result in a
reconstruction close to the original image
4 EXPERIMENT AND RESULTS
To evaluate the adequacy of the proposed
architecture, we conducted an experiment using the
Orca dataset and compared the results with other
papers that employ GAN approaches to generate
CECT images from NECT images..
The network hyperparameters were set as
follows: 100 epochs, the Adam optimizer with beta1
= 0.5 and beta2 = 0.9, a learning rate of 10^-4 for the
diffusion method and GAN, a batch size of 2, T =
1000, which represents the number of interactions in
the noising and denoising process, a step size of k =
250, and T/k = 4 diffusion steps. The weight loss in
diffusion and cycle models was set to λ1φ = 0.5
Diffusion Model for Generating Synthetic Contrast Enhanced CT from Non-Enhanced Heart Axial CT Images
861
The metrics were obtained through the
comparison between generated CECT images with
real ones. The metrics presented in Table 3 are based
on the averages of these results.
The results obtained, exemplified by Figure 2 and
Table 3, showcase the remarkable performance of the
proposed diffusion model. Notably, the
competitiveness of the PSNR and SSIM indicators in
generating contrast-enhanced heart images reflects
the model's significant ability to preserve both quality
and visual similarity.
Figure 2: Images with contrast generated by the network
from non-contrast images.
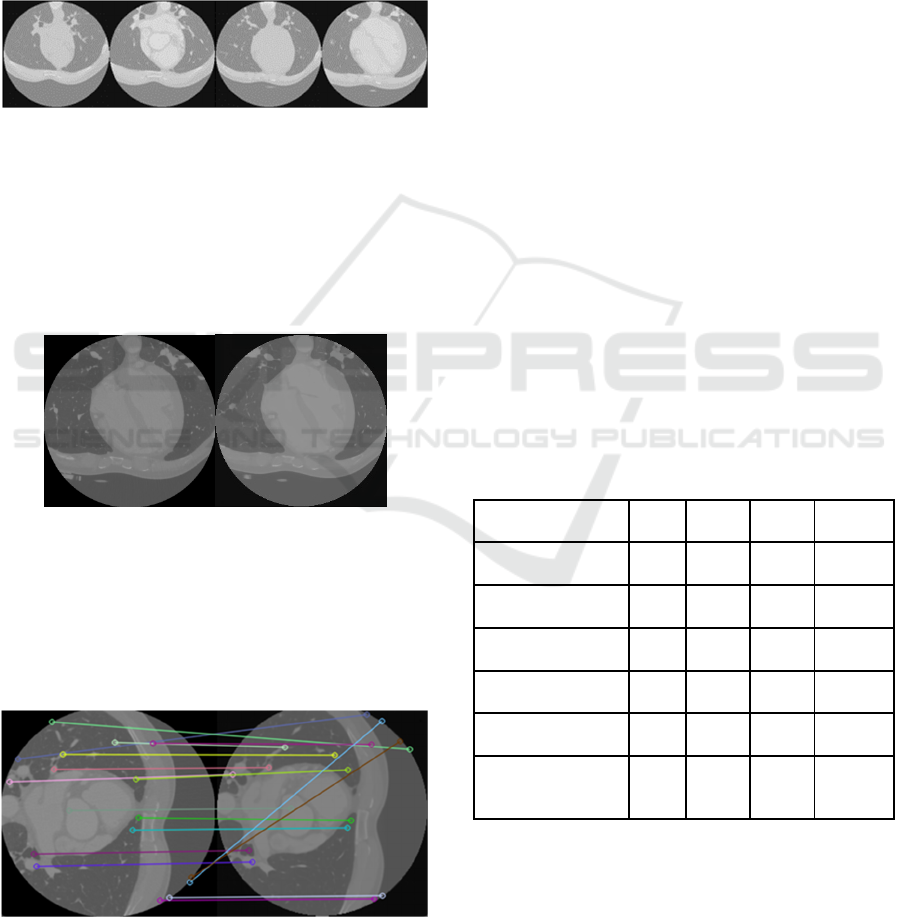
A noteworthy point is that although the images
have a high degree of visual similarity, MAE and
RMSE values are still much higher than expected. A
good example is the two images below, which exhibit
considerable visual resemblance but yield MAE and
RMSE values as high as 0.6 and 0.7, respectively.
Figure 3: Real Contrast Images (Left) and the Generated
One (Right).
However, in other images, the MAE and RMSE
values reached 0.11 and 0.15, respectively,
demonstrating that depending on the image, the
network can generate a more accurate version closer
to the real one.
Figure 4: A Feature Matching of Real Contrast Images
(Left) and the Generated One (Right).
However, upon analysing the Root Mean Square
Error (RMSE) values, it is observed that, despite the
visual resemblance of the generated images, the
model predictions deviate significantly from the
actual values, as seen in Table 3.
Upon closer examination of the images, a subtle
yet discernible variance in the absolute pixel values
between the original and the generated samples
becomes apparent. Indeed, a slight disparity exists
between the generated pixel values and the
corresponding ideal pixel values, as shown in Figure
2, with the generated pixel values exhibiting a
marginally higher magnitude. While these minor
discrepancies may seem inconspicuous individually,
their cumulative effect in the summation process
significantly contributes to the observed dissimilarity
reflected in the RMSE.
It is important to note that the interpretation of
RMSE depends on the specific domain of the problem
and the units of the variable being predicted. In some
cases, a high RMSE may be acceptable if it aligns
with the natural variations in the data or is justified by
the nature of the problem being addressed. These
indicators suggest the presence of substantial
variations that require a deeper understanding of the
generated images. Studying these discrepancies can
provide valuable insights further to enhance the
effectiveness of the image generation process.
Table 3: Comparison of results among CyTran, Cycle-
GAN, Pix2Pix-GAN networks, Cycle-GAN-2D, and
Cycle-GAN-2D with SkipResidual Generator against the
proposed model.
Model MAE RMSE SSIM PSRN
CyTran 0.061 0.144 0.745 29.66
C
y
cle-GAN 0.066 0.150 0.724 29.22
Pix2Pix-GAN 0.070 0.165 0.729 29.51
C
y
cle-GAN-2D 0,030 - 0,433 15,569
Pix2Pix-GAN-2D 0,025 - 0,492 16,375
Proposed
Diff-Model
0.061 0.200 0.701 32,85
5 CONCLUSIONS
This paper addresses the translation of contrast and
non-contrast cardiac computed tomography (CT)
images using a deep learning-based adversarial
ICEIS 2024 - 26th International Conference on Enterprise Information Systems
862

diffusion model. By overcoming challenges
associated with medical image translation, we
explore an approach that combines concepts from
generative adversarial networks (GANs) and
diffusion models. The obtained results, evaluated
through metrics such as PSRN and SSIM, showcase
the remarkable capability of the model in generating
contrast-enhanced cardiac images while preserving
quality and visual similarity. However, the analysis of
RMSE indicates persistent challenges, suggesting the
presence of variations that require a deeper
understanding to enhance the consistency and fidelity
of the generated images.
In conclusion, the developed model delivers
notable results, but the study acknowledges the need
for continuous improvements to address variations in
the generated images. The intersection of GANs and
diffusion models proves promising, pointing towards
future research and developments in medical image
translation and significantly contributing to
advancing this crucial area in clinical practice.
ACKNOWLEDGEMENTS
The authors acknowledge the Coordenação de
Aperfeiçoamento de Pessoal de Nível Superior
(CAPES), Brazil - Finance Code 001, Conselho
Nacional de Desenvolvimento Científico e
Tecnológico (CNPq), Brazil, and Fundação de
Amparo à Pesquisa Desenvolvimento Científico e
Tecnológico do Maranhão (FAPEMA) (Brazil),
Empresa Brasileira de Serviços Hospitalares (Ebserh)
Brazil (Grant number 409593/2021-4), and the
Portuguese funding agency, FCT - Fundação para a
Ciência e a Tecnologia, within project
UIDB/50014/2020.DOI.10.54499/UIDB/50014/202
0 | https://doi.org/10.54499/uidb/50014/2020 for the
financial support.
REFERENCES
Azarfar, G., Ko, SB., Adams, S.J. et al. (2023) Applications
of deep learning to reduce the need for iodinated
contrast media for CT imaging: a systematic review. Int
J CARS 18, 1903–1914. https://doi.org/10.1007/s115
48-023-02862-w
Choi, J.W., Cho, Y.J., Ha, J.Y. et al. (2021) Generating
synthetic contrast enhancement from non-contrast chest
computed tomography using a generative adversarial
network. Sci Rep 11, 20403.
https://doi.org/10.1038/s41598-021-00058-3
Chun, J., Chang, J. S., Oh, C., Park, I., Choi, M. S., Hong,
C. S., ... & Kim, J. S. (2022). Synthetic contrast-
enhanced computed tomography generation using a
deep convolutional neural network for cardiac
substructure delineation in breast cancer radiation
therapy: a feasibility study. Radiation Oncology, 17(1),
1-9.
Corballis, N.; Tsampasian, V.; Merinopoulis, I.;
Gunawardena, T.; Bhalraam, U.; Eccleshall, S.; Dweck,
M.R.; Vassiliou, V. CT.(2023) angiography compared
to invasive angiography for stable coronary disease as
predictors of major adverse cardiovascular events—A
systematic review and meta-analysis. Heart Lung, 57,
207–213.
Counseller, Q. and Aboelkassem, Y. (2023) Recent
technologies in cardiac imaging, Frontiers in Medical
Technology, Vol. 4, DOI 10.3389/fmedt.2022.984492,
Croitoru, F. A., Hondru, V., Ionescu, R. T., & Shah, M.
(2023). Diffusion Models in Vision: A Survey, in IEEE
Transactions on Pattern Analysis and Machine
Intelligence, vol. 45, no. 9, pp. 10850-10869.
Dondi M, Paez D, Raggi P, Shaw LJ, Vannan
M.(2021).Integrated non-invasive cardiovascular
imaging: a guide for the practitioner. International
Atomic Energy Agency.
Domingues, R. A. D. (2022). Automatic contrast generation
from contrastless CTs, Master Thesis, Universidade do
Porto, FCUP - Faculdade de Ciências.
Goodfellow, I., Pouget-Abadie, J., Mirza, M., Xu, B.,
Warde-Farley, D., Ozair, S., ... & Bengio, Y. (2020).
Generative adversarial networks. Communications of
the ACM, 63(11), 139-144.
H., & Kwak, S. (2021). Neural contrast enhancement of CT
image. In Proceedings of the IEEE/CVF Winter
Conference on Applications of Computer Vision (pp.
3973-3982).
Henry, J., Natalie, T., & Madsen, D. (2021). Pix2Pix GAN
for Image-to-Image Translation. Research Gate
Publication, 1-5.
Ho, J., Jain, A., & Abbeel, P. (2020). Denoising diffusion
probabilistic models. Advances in neural information
processing systems. Editors: H. Larochelle and M.
Ranzato and R. Hadsell and M.F. Balcan and H. Lin},
Vol. 33, pages 6840--685}, Curran Associates, Inc.},
https://proceedings.neurips.cc/paper_files/paper/2020
/file/4c5bcfec8584af0d967f1ab10179ca4b-Paper.pdf
Hu, T., Oda, M., Hayashi, Y., Lu, Z., Kumamaru, K. K.,
Akashi, T., ... & Mori, K. (2022). Aorta-aware GAN for
non-contrast to artery contrasted CT translation and its
application to abdominal aortic aneurysm detection.
International Journal of Computer Assisted Radiology
and Surgery, 1-9. https://doi.org/10.1007/s11548-021-
02492-0
Jing, Y., Yang, Y., Feng, Z., Ye, J., Yu, Y., & Song, M.
(2019). Neural style transfer: A review. IEEE
transactions on visualization and computer graphics,
26(11), 3365-3385.
Karras, T., Aittala, M., Aila, T., & Laine, S. (2022).
Elucidating the design space of diffusion-based
generative models. Advances in Neural Information
Processing Systems, 35, 26565-26577.
Diffusion Model for Generating Synthetic Contrast Enhanced CT from Non-Enhanced Heart Axial CT Images
863

Park, T., Liu, M. Y., Wang, T. C., & Zhu, J. Y. (2019).
Semantic image synthesis with spatially-adaptive
normalization. In Proceedings of the IEEE/CVF
conference on computer vision and pattern recognition
(pp. 2337-2346).
Parmar, G., et all. (2023). Zero-shot Image-to-Image
Translation. In ACM SIGGRAPH 2023 Conference
Proceedings (SIGGRAPH '23). Association for
Computing Machinery, New York, NY, USA, Article
11, 1–11. https://doi.org/10.1145/3588432.3591513
Peebles, W. and Xie, S. (2023) Scalable Diffusion Models
with Transformers," IEEE/CVF International
Conference on Computer Vision (ICCV), Paris, France,
2023, pp. 4172-4182, doi: 10.1109/ICCV51070.20
23.00387
Radford, A., Metz, L., & Chintala, S. (2015). Unsupervised
representation learning with deep convolutional
generative adversarial networks. arXiv preprint
arXiv:1511.06434.
Ronneberger, O., Fischer, P., & Brox, T. (2015). U-net:
Convolutional networks for biomedical image
segmentation. In Medical Image Computing and
Computer-Assisted Intervention–MICCAI 2015: 18th
International Conference, Munich, Germany, October
5-9, 2015, Proceedings, Part III 18 (pp. 234-241).
Springer International Publishing.
Ristea, N. C., Miron, A. I., Savencu, O., Georgescu, M. I.,
Verga, N., Khan, F. S., & Ionescu, R. T. (2021). Cytran:
Cycle-consistent transformers for non-contrast to
contrast ct translation. arXiv preprint arXiv:2110.064
00.
Seo, M. et al., "Neural Contrast Enhancement of CT
Image", 2021 IEEE Winter Conference on Applications
of Computer Vision (WACV), Waikoloa, HI, USA,
2021, pp. 3972-3981, doi: 10.1109/WACV48630.20
21.00402.
Özbey, M., Dalmaz, O., Dar, S. U., Bedel, H. A., Özturk,
Ş., Güngör, A., & Çukur, T. (2023). Unsupervised
medical image translation with adversarial diffusion
models. IEEE Transactions on Medical Imaging.
Wang, Z., Zheng, H., He, P., Chen, W., & Zhou, M. (2022).
Diffusion-gan: Training gans with diffusion. arXiv
preprint arXiv:2206.02262.
Wolterink, J., Vo, B.D., Leine, T. , Viergever, M. A.,
Išgum, I.(2022) Orca score. https://orcascore.grand-
challenge.org
WHO World Health Organization. Noncommunicable
diseases. (2023) Available online: https://www.who.
int/news-room/fact-sheets/detail/noncommunicable-
diseases.
Zhu, J. Y., Isola, P .Zhou, T., & Efros, A. A. (2017). Image-
to-image translation with conditional adversarial
networks. In Proceedings of the IEEE conference on
computer vision and pattern recognition (pp. 1125-
1134).
ICEIS 2024 - 26th International Conference on Enterprise Information Systems
864
